Spermatocytic Tumor of Testis in a Young Male
By Madiha Bilal Qureshi, Nasir Uddin, Zubair Ahmad, Saira FatimaAffiliations
doi: 10.29271/jcpsp.2021.06.722ABSTRACT
Spermatocytic tumor is a rare germ cell tumor not related to germ cell neoplasia in situ, and derived from postpubertal-type germ cells. It was previously called “Spermatocytic Seminoma” due to false belief of its origin from germ cell neoplasia in situ. The tumor usually occurs in an older age group and orchidectomy is curative. We present a case of spermatocytic tumor in a 25-year male who presented with right-sided testicular swelling and right-sided varicocele. Radiology revealed a 9.8 × 9 cm testicular mass and the patient underwent right-sided orchidectomy. Microscopic examination showed classic morphology with three characteristic cell types and diagnosis of spermatocytic tumor was made.
Key Words: Spermatocytic tumor, testis, young.
INTRODUCTION
Spermatocytic tumor accounts for 1% of testicular germ cell tumors originating from post-pubertal type germ cells.1 The tumor arises only in descended testis. It usually occurs in the fifth to sixth decades of life, an age group much greater compared to that of germ cell tumors derived from germ cell neoplasia in situ.2 About 10% are bilateral. Clinically, it appears as a round painless swelling of long duration.3 Radiography demonstrates a well-circumscribed heterogenous mass. Gross features include a soft, mucoidy, gelatinous, friable cut surface often showing cystic spaces. Microscopically, spermatocytic tumor reveals three distinct intermixed populations of cells, which are negative for immunostain placental alkaline phosphatase (PLAP) with absence of intracytoplasmic glycogen and positivity for immunostain cluster of differentiation (CD117).4 Prognosis is excellent with rare cases exhibiting sarcomatous transformation and distant metastasis.5
We, herein, present a case of a 25-year male with painless testicular swelling for three years along with right-sided varicocele.
CASE REPORT
A 25-year male presented with history of painless right testicular swelling for three years along with right-sided varicocele. Ultrasound showed a 9.8 × 9 cm testicular swelling with varicocele.
The specimen received was coded as “Right testicular mass”. It was a testis measuring 16.5 × 14.5 × 9.5 cm with spermatic cord that measured 6.5 × 3 cm. The tunica albuginea and vaginalis were intact. The specimen weighed 645 grams. Cut surface revealed a tan to pale lobular soft fleshy lesion measuring 12.5 × 10.5 cm limited to the testis.
Microscopy showed a tumor composed of diffuse pattern of three types of cells: small cells with round, uniform, dense hyperchromatic nuclei and scant cytoplasm; intermediate cells with finely granular to filamentous chromatin and variably- prominent nucleoli; and giant cells with eosinophilic to amphophilic cytoplasm, single to multiple nuclei and spireme chromatin (Figures 1 and 2). Focal cystic change was seen. The stroma was fibrous and brisk mitotic figures including atypical mitoses were appreciated. Tumor lacked lymphocytic infiltrate and areas of high grade spindle cell or pleomorphic cell proliferation and necrosis. The cells were CD117 positive, PLAP and leucocyte common antigen (LCA) negative. Intracytoplasmic glycogen was absent. Based on these combined features, it was diagnosed as spermatocytic tumor.
DISCUSSION
Spermatocytic tumor is a redefined rare entity derived from postpubertal-type germ cells.1 It accounts for 1% of all testicular tumors with a mean patient age of 52 – 59 years.2 Its incidence peaks in the sixth decade of life, usually in white men; but in this case, the patient was of 25 years and non-Caucasion.3 The tumor occurs exclusively in testis and lacks extra-testicular or female counterpart; and is not associated with cryptorchidism, unlike other germ cell tumors. Age-related activating mutations in the FGFR3 and HRAS genes and amplification of the short arm of chromosome 9 in spermatogonia are suggested etiologic factors.6
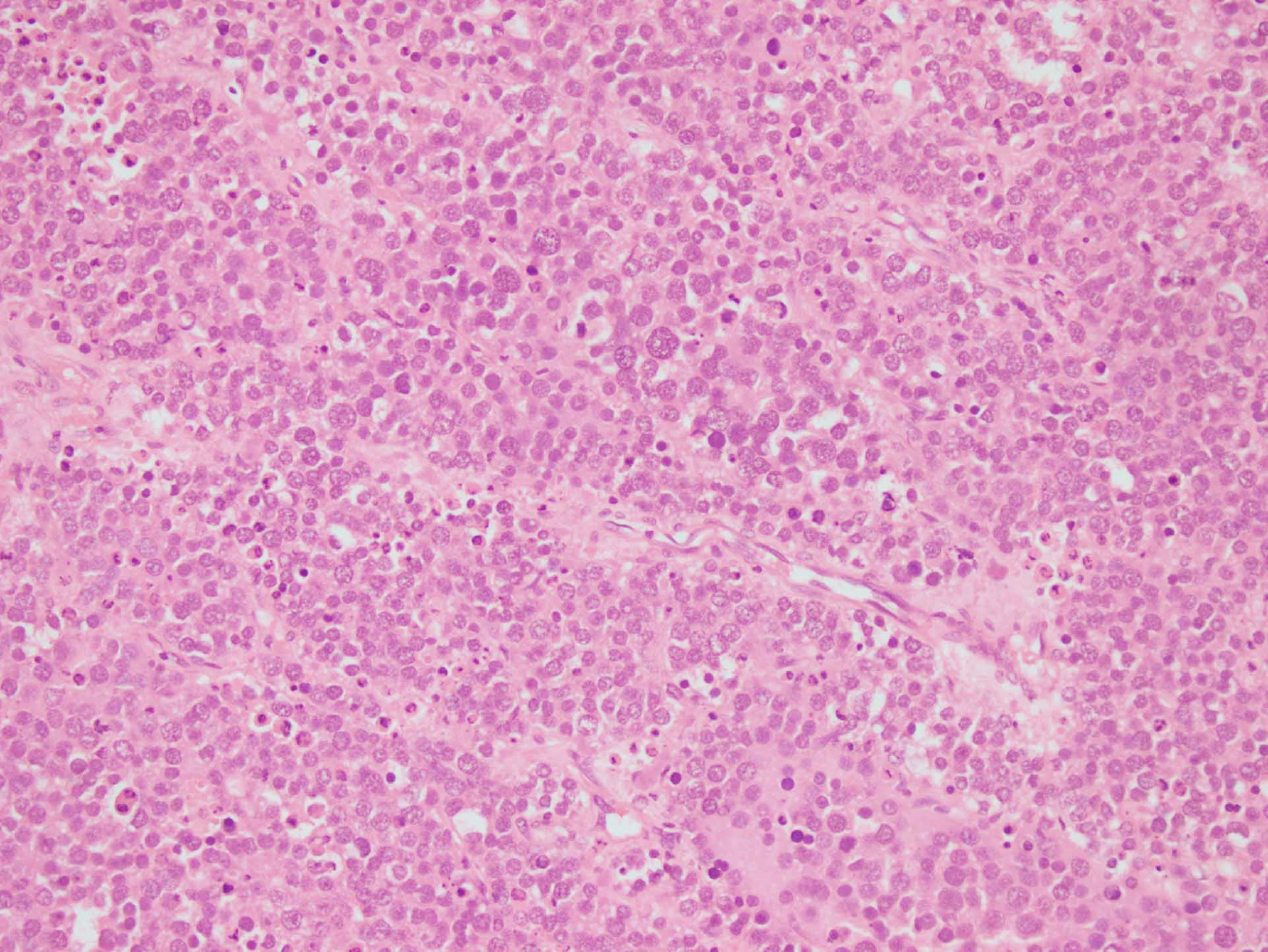 Figure 1: Diffuse sheets of variable-sized tumor cells separated by thin fibrous septae (H&E, 4x).
Figure 1: Diffuse sheets of variable-sized tumor cells separated by thin fibrous septae (H&E, 4x).
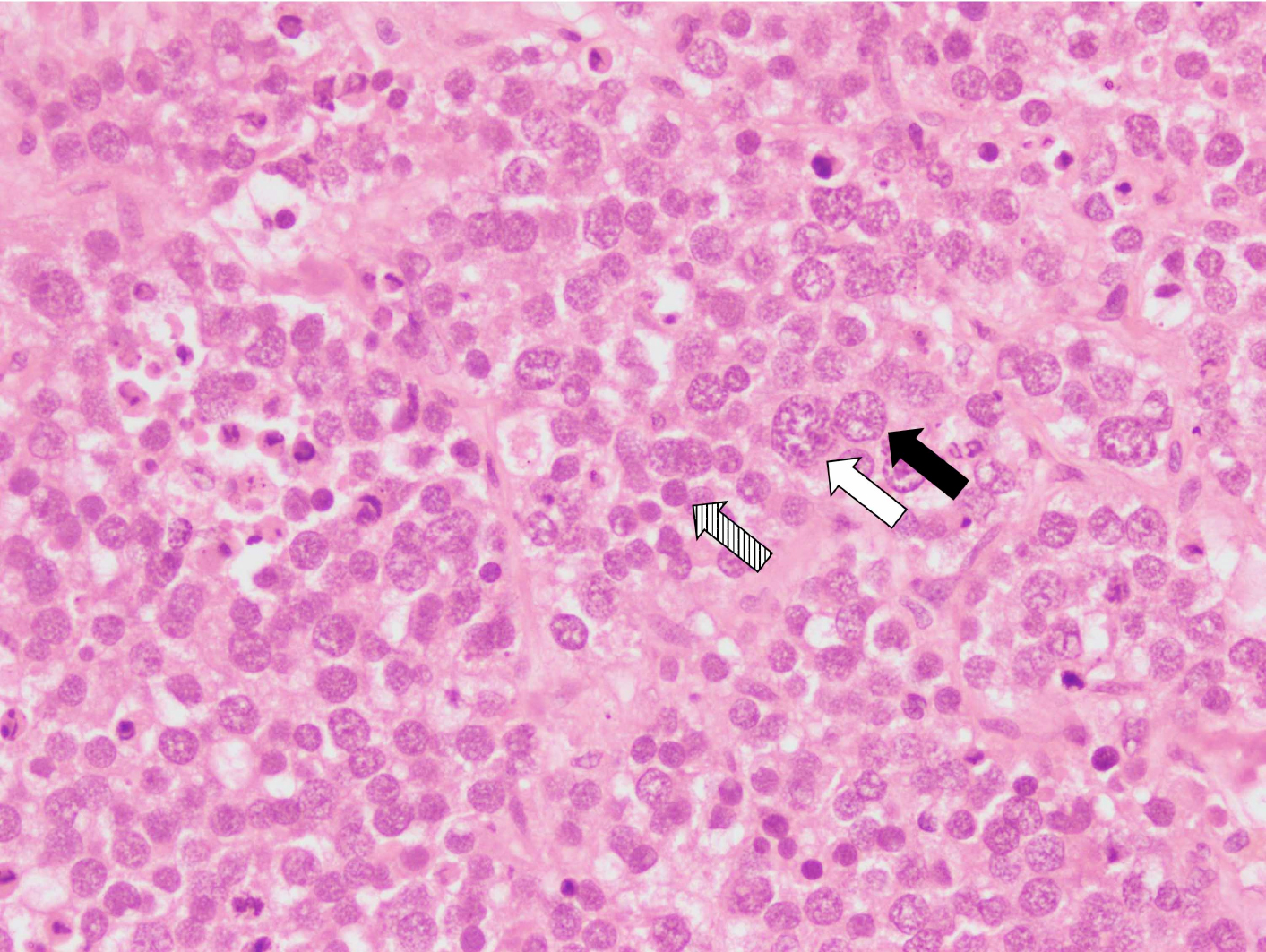 Figure 2: High-power view shows giant cells (white arrow), and intermediate cells (black arrow) with characteristic spireme chromatin. Striped white arrow shows small lymphocyte-like cells with scant cytoplasm and hyperchromatic nuclei. (H&E, 20x).
Figure 2: High-power view shows giant cells (white arrow), and intermediate cells (black arrow) with characteristic spireme chromatin. Striped white arrow shows small lymphocyte-like cells with scant cytoplasm and hyperchromatic nuclei. (H&E, 20x).
It usually presents as a painless testicular mass with bilateral involvement in 10% of the cases. Serum markers are not raised. Prognosis is excellent with rare metastasis unless complicated by sarcoma; and orchidectomy stays the treatment unlike surgery and chemoradiation in other germ cell tumors.7 The tumor ranges in size from 3-5 cm and has a multinodular configuration with rare extension into paratesticular tissue. Cut surface has a variegated appearance including grey white, haemorrhagic, fleshy, edematous or myxoid areas. Microscopy shows a diffuse pattern of non-cohesive tumor cells of three types: small cells with round, uniform dense hyperchromatic nuclei and scant cytoplasm resembling lymphocytes; intermediate-sized cells (predominant type) with thread-like chromatin and variably prominent nucleoli; and giant cells with moderate cytoplasm, single to multiple nuclei and spireme chromatin.8 The stroma is usually scant, finely fibrous with edema and cystic change. Lymphocytic infiltration is mild to absent. Brisk mitotic figures including atypical mitoses are seen. Vascular, tunical and epididymal invasion can be present; however, granulomatous inflammation is rare.9 Intratubular gem cell neoplasia is absent (Table I).
Table I: Comparison of spermatocytic tumor with classic seminoma.
|
Tumor type |
Spermatocytic tumor |
Classic seminoma |
|
Proportion |
1-2% |
40-50% |
|
Bilaterality |
9% |
2% |
|
Median age |
52-59 years |
40 years |
|
Association with germ cell neoplasia In situ |
No |
Yes |
|
Site |
Testis only |
Testis, mediastinum, pineal gland, retroperitoneum |
|
Composition |
3 cell types |
1 cell type |
|
Lymphoid reaction |
Rare to absent |
Present |
|
Stroma |
Scanty |
Prominent |
|
Sarcomatoid transformation |
Occasional |
Absent |
Immunostaining is helpful in establishing the diagnosis. Tumor is negative for the usual embryonic germ cell markers including OCT 3/4, PLAP, AFP, Beta hCG and CD30 and positive for proteins MAGEA4, KIT, SAGE1, DMRT1, SALL4, OCT2 and synovial sarcoma X breakpoint proteins.10,11 Intracytoplasmic glycogen is absent. Vascular invasion, in addition to sarcoma component, is a poor prognostic factor.12
In conclusion, spermatocytic tumor is a rare testicular tumor with extremely rare occurrence in the young. It has a characteristic histologic appearance and correct diagnosis may have great impact on treatment and prognosis.
PATIENT’S CONSENT:
Informed consent has been taken from the patient.
CONFLICT OF INTEREST:
The authors declared no conflict of interest.
AUTHORS’ CONTRIBUTION:
MBQ: Drafted the case summary and discussion, and edited the manuscript.
NU, ZA, SF: Established and confirmed the diagnosis, drafted the manuscript.
REFERENCES
- Lombardi M, Valli M, Brisigotti M, Rosai J. Spermatocytic seminoma: Review of the literature and description of a new case of the anaplastic variant. Int J Surg Pathol 2011; 19(1):5-10. doi: 10.1177/1066896910388645.
- Carriere P, Baade P, Fritschi L. Population-based incidence and age distribution of spermatocytic seminoma. J Urol 2007; 178(1):125-8. doi: 10.1016/j.juro.2007.03.024.
- Decaussin M, Borda A, Bouvier R, Ruffion A, David C, Agard C, et al. Spermatocytic seminoma: A clinicopathological and immunohistochemical study of 7 cases. Ann Pathol 2004; 24(2):161-6. doi: 10.1016/s0242-6498(04)93939-9.
- Cummings OW, Ulbright TM, Eble JN, Roth LM. Spermato-cytic seminoma: An immunohistochemical study. Hum Pathol 1994; 25(1):54-9. doi: 10.1016/0046-8177(94)90171-6.
- Albores Saavedra J, Huffman H, Alvarado Cabrero I, Ayala AG. Anaplastic variant of spermatocytic seminoma. Hum Pathol 1996; 27(7):650-5. doi: 10.1016/s0046-8177(96) 90393-7.
- Reuter VE. Origins and molecular biology of testicular germ cell tumors. Mod Pathol 2005; 18(S2):S51. doi: 10.1038/ modpathol.3800309.
- Steiner H, Gozzi C, Verdorfer I, Mikuz G, Bartsch G, Hobisch A. Metastatic spermatocytic seminoma-an extremely rare disease. Eur Uro 2006; 49(1):183-6. doi: 10.1016/j. eururo.2005.08.020.
- Burke AP, Mostofi FK. Spermatocytic seminoma: A clinico-pathologic study of 79 cases. J Urol Pathol 1993; 1(6): 21-32.
- Haroon S, Tariq MU, Fatima S, Kayani N. Spermatocytic seminoma: A 21 year’s retrospective study in a tertiary care hospital in Pakistan. Int J Clin Exp Patho 2013; 6(11):2350.
- Cao D, Li J, Guo CC, Allan RW, Humphrey PA. SALL4 is a novel diagnostic marker for testicular germ cell tumors. Am J Surg Pathol 2009; 33(7):1065-77. doi: 10.1097/PAS. 0b013e3181a13eef.
- Verdorfer I, Rogatsch H, Tzankov A, Steiner H, Mikuz G. Molecular cytogenetic analysis of human spermatocytic seminomas. J Pathol. 2004; 204(3):277-81. doi: 10.1002/ path.1634.
- Menon S, Karpate A, Desai S. Spermatocytic seminoma with rhabdomyosarcomatous differentiation: A case report with a review of the literature. J Can Res Ther 2009; 5(3):213. doi: 10.4103/0973-1482.57131.