Radiologic and Histopathologic Features of a Neck-Localised Follicular Dendritic Cell Sarcoma: A Case Report and Literature Review
By Jing Zeng, Xi Xu, Changzheng Shi, Liangping LuoAffiliations
doi: 10.29271/jcpsp.2022.05.674ABSTRACT
Follicular dendritic cell sarcoma (FDCS) is an uncommon tumour. The pathological features of FDCS have been extensively described in the literature, but there are few reports on its imaging features. A 48-year-old man presented at our hospital with a mass in the left submandibular region without associated oral complaints. Contrast-enhanced magnetic resonance imaging revealed a mass with smooth margins, and postoperative histopathological and immunohistochemical findings were consistent with FDCS. At present, our understanding of FDCS is still insufficient, and its diagnosis is dependent on pathological evaluation. We aim to provide a bit of information for clinicians to consider when they see these types of tumours.
Key Words: Follicular dendritic cell sarcoma, Magnetic resonance imaging, Imaging features, Histopathology.
INTRODUCTION
Follicular dendritic cell sarcoma (FDCS) is a rare tumor and exhibits an unpredictable clinical course.1 FDCS is a neoplastic lesion of follicular dendritic cells (FDC)2, which are constituent parts of B cell follicles in secondary lymphoid organs and originate from mesenchymal cells.1 FDCS predominantly involves lymph nodes, particularly the cervical and axillary lymph nodes, and extranodal cases are rare.3 Herein, we report the magnetic resonance imaging (MRI) findings of a case of FDCS in the left submandibular space in the upper neck and review the related literature.
CASE REPORT
A 48-year-old man was admitted to our hospital in April 2018 with a two-year history of painless swelling in the left submandibular region. He reported no associated oral or systemic complaints. A well-defined, firm, non-tender neoplasm approximately 6×8 cm in size was palpated in his left submandibular region. Laboratory investigations showed slightly elevated monocytes (10.37%).
Further evaluation was done with MRI, which showed a round, well-circumscribed, 3.7×3.9×5.1 cm mass, adjacent to the left submandibular gland that was compressing adjacent structures (Figure 1 A,B,E). The mass showed obvious restricted diffusion on diffusion-weighted imaging (Figure 1C). After administration of intravascular contrast, the mass showed mild enhancement with some hypointense spotty areas (Figure 1D, 1F). Several enlarged lymph nodes were found around the lesion. Based on MRI findings, the diagnosis was made of a malignant tumour with lymphatic metastasis.
An excision biopsy was performed. Microscopically, spindle and ovoid tumour cells were arranged in swirls or sheets (Figure 2A, 2B), and the normal structure of the lymph node was effaced, which demonstrated that the neoplasm occurred in the lymph node. There were >40 mitoses per 10 high power fields. Positive staining for CD21 (Figure 2C), Vimentin (Figure 2D), and CD68 (not shown) were observed. The rate of positive expression of P53 was 5% and Ki-67 was 30%. A final histopathological diagnosis of FDCS was made. The patient was in good condition at the 3-month follow-up.
DISCUSSION
FDCS, with a risk of local recurrence (nearly 40% to 50%) and metastasis, behaves as an intermediate grade sarcoma.2,4 FDCS mainly affects young to middle-aged people with no gender differences.2 It can occur in different parts of the body where it can mimic various tumours and even inflammatory processes.1 A slowly growing, symptomless, painless cervical lymphadenopathy is the most common manifestation.4
Table I: Imaging performances of 4 previously reported cases and ours of follicular dendritic cell sarcoma.
|
Study |
Imaging tests |
Sex |
Age (years) |
Location |
Sizes (cm) |
Borders |
Imaging findings |
|
Qiu et al. |
CT |
Man |
38 |
Right cervical lymph node |
4.5 (diameter) |
Well-delineated |
Homogeneous soft-tissue attenuation |
|
Enhanced- CT |
Woman |
35 |
Left cervical lymph nodes |
1.5-3 (diameter) |
Well-delineated |
Homogenous moderate enhancement |
|
|
McClelland et al. |
MRI |
Woman |
51 |
Right parotid tail |
2.5 (diameter) |
Relatively well-delineated |
Hyperintense in fat-suppressed T2-weighted imaging and relatively homogeneous |
|
Singer et al. |
Enhanced- CT |
Man |
60 |
Along the right anterior cervical lymph node chain |
7.4×7.8× 6.5 |
Well-delineated |
Heterogeneous density; Mildly heterogeneous enhancement (predominantly capsular enhancement) |
|
This case |
Enhanced- MRI |
Man |
48 |
In the left submandibular space |
3.7×3.9× 5.1 |
Well-delineated |
Hypointense on T1-weighted imaging and slightly heterogeneous hyperintense on T2-weighted imaging; Mild enhancement |
|
CT = Computed tomography; MRI = Magnetic resonance imaging. |
|||||||
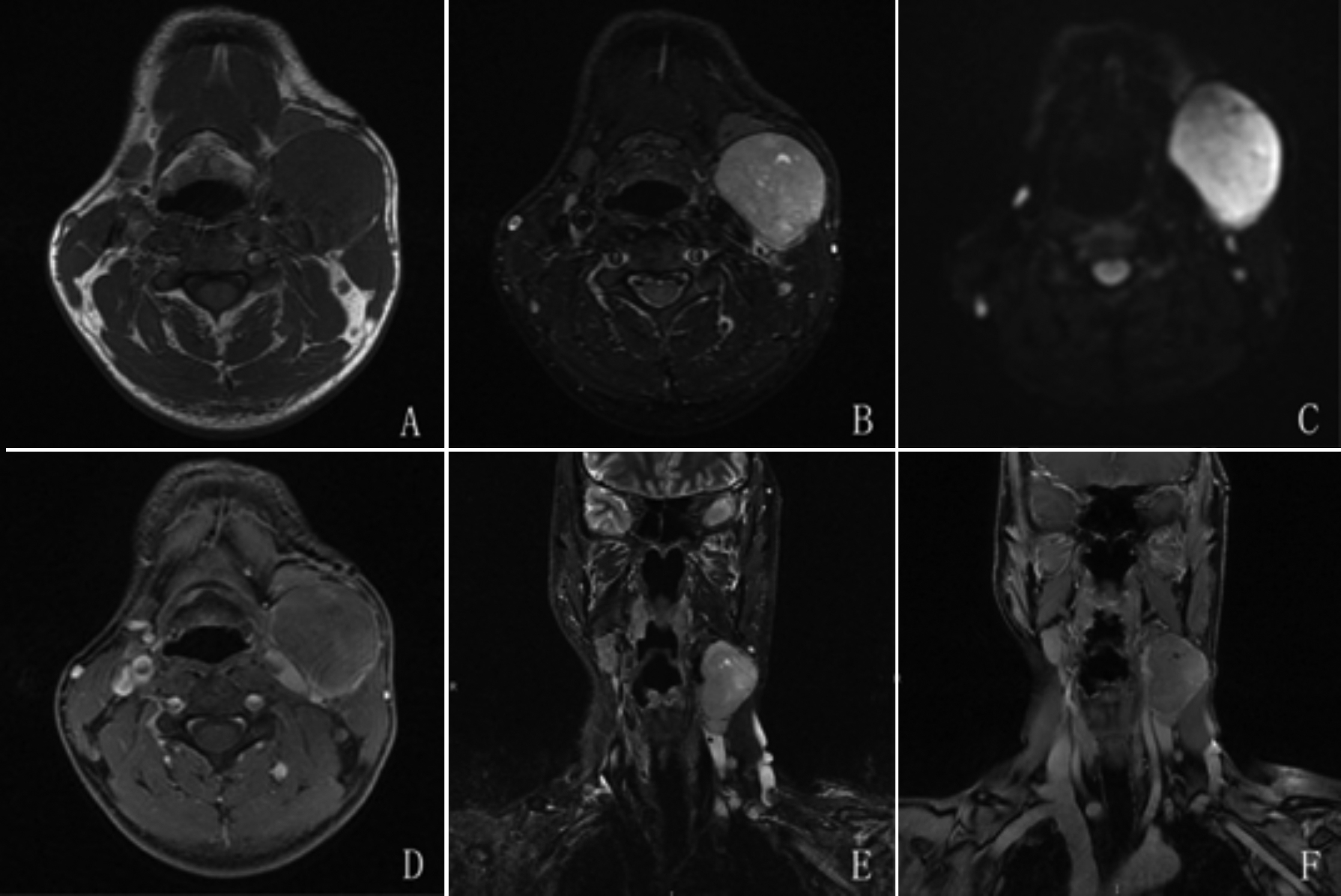 Figure 1: Magnetic resonance imaging (MRI) findings of the case. A mass is seen adjacent to the left submandibular gland and lateral to the external jugular vein. The oval-shaped and well-circumscribed mass was noted to be hypointense on T1-weighted imaging (A) and hyperintense on T2-weighted imaging with fat suppression (B,E). The mass was hyperintense on diffusion-weighted imaging (C) with mild enhancement (D,F).
Figure 1: Magnetic resonance imaging (MRI) findings of the case. A mass is seen adjacent to the left submandibular gland and lateral to the external jugular vein. The oval-shaped and well-circumscribed mass was noted to be hypointense on T1-weighted imaging (A) and hyperintense on T2-weighted imaging with fat suppression (B,E). The mass was hyperintense on diffusion-weighted imaging (C) with mild enhancement (D,F).
Previously, the radiologic features have not been well described due to the rarity of this lesion. The imaging findings of FDCS in mediastinum on computed tomography (CT) were firstly reported by Leipsic et al.,5 showing a soft tissue density lesion with lumpy calcification. They suggested that two potential features of FDCS were massive calcification, less central necrosis, and hemorrhage in large tumours.5 In four cases reported by Long-Hua et al.,6 Singer et al.7 and McClelland et al.,8 FDCS presented in the head-and-neck region. A summary of the imaging features of these cases and our case is shown in Table I. The aforementioned cases of FDCS in the head and neck region share some imaging characteristics with our case, such as unilaterality, well-circumscribed boundaries, and compression of adjacent structures. Cases in which enhanced CT or MRI were performed displayed mild to moderate enhancement. Necrosis was noted only in our case and in the case of Singer et al.,7 possibly because the tumours in the other cases were small.
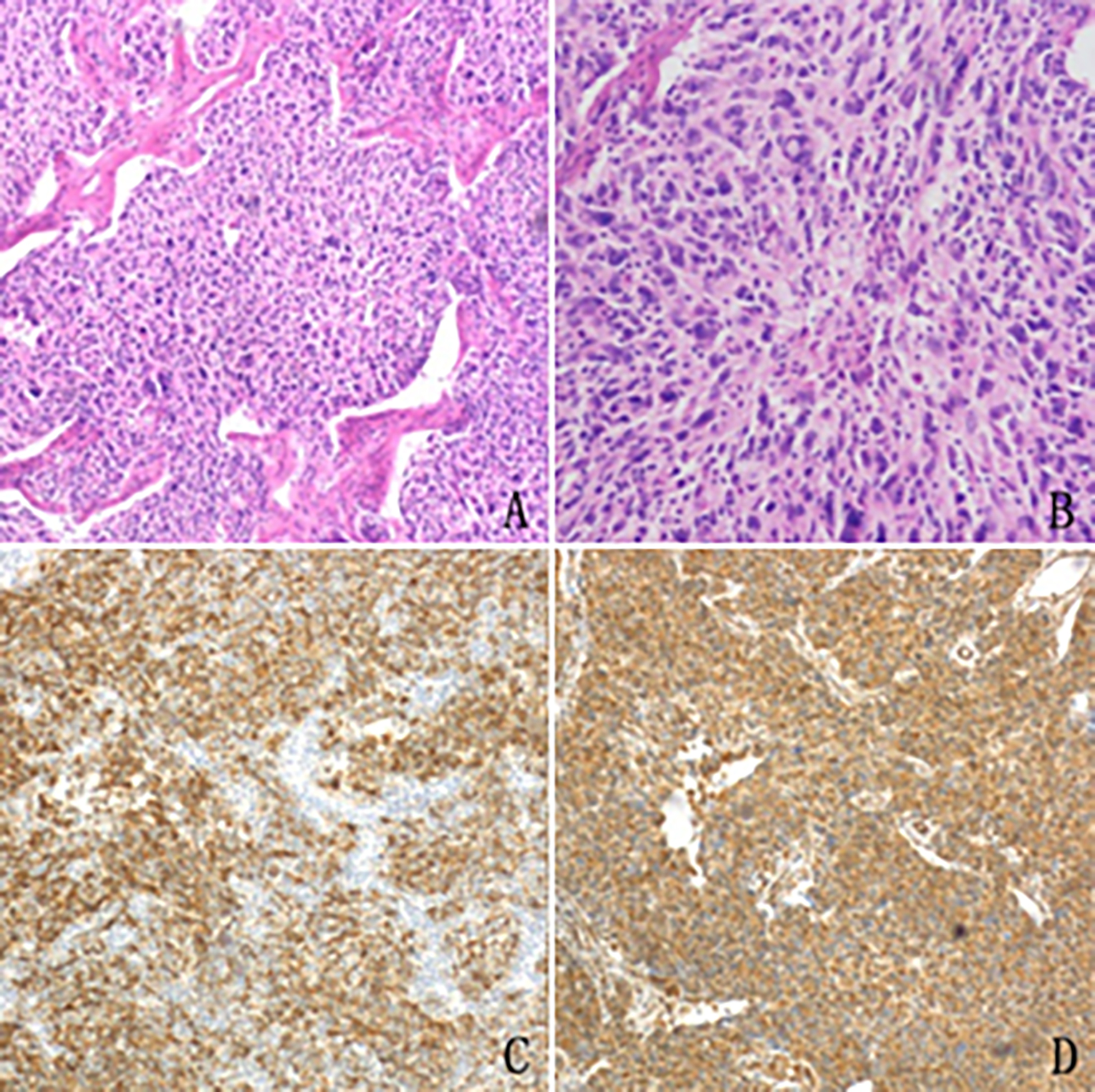 Figure 2: Histopathological features of the excised mass in the case. (A,B) Spindle, round or ovoid tumour cells are arranged in a storiform pattern with whorled architecture (H and E stain, original magnification, ×40 and ×100). Strong positive staining for CD21 (C) and Vimentin (D) is observed.
Figure 2: Histopathological features of the excised mass in the case. (A,B) Spindle, round or ovoid tumour cells are arranged in a storiform pattern with whorled architecture (H and E stain, original magnification, ×40 and ×100). Strong positive staining for CD21 (C) and Vimentin (D) is observed.
Differential diagnosis of FDCS in the head-and-neck region should include schwannoma, head and neck squamous cell carcinoma, and lymphadenopathy such as lymphoma or lymph-node metastatic tumours.7 Both schwannoma and lymph-node metastatic tumours often show central necrosis, while multiple areas are often involved in lymphoma. Head and neck squamous cell carcinomas generally present with invasive boundaries such as infiltration into the adjacent fat or muscle; therefore, in our case, a squamous cell carcinoma was ruled out as the tumour had a well-circumscribed margin and no adjacent tissue infiltration was observed.
Histopathological evaluation is the mainstay of diagnosing FDCS. The spindly, ovoid tumour cells characteristically form a fascicular, or diffuse sheet-like pattern under light microscope.9 Immunohistochemical staining is also essential and markers such as CD23, CD35, CD21 have been widely used.3
In comparison with other treatments, early-diagnosed cases of FDCS treated with surgical removal had higher overall survival (OS); therefore, the main therapy for such patients should be surgery.4
Based on our observations, we suggest that the diagnostically useful imaging features of FDCS in the head and neck region are unilaterality and circumscription with mild to moderate enhancement and paucity of necrosis in relatively small tumours. We hope our data will inform clinicians to keep this tumor in the differential diagnoses when encountering these types of tumours.
PATIENT’S CONSENT:
The patient’s consent for publication of fully anonymised imaging was waived.
ACKNOWLEDGEMENT:
This work was supported by the Guangzhou Key Laboratory of Molecular and Functional Imaging for Clinical Translation (Project No. 201905010003).
COMPETING INTEREST:
The authors declared no conflict of interest.
AUTHORS’ CONTRIBUTION:
JZ: Design of the study, data collection, literature review and manuscript writing.
XX: Literature review, imaging processing.
CS: Literature review and manuscript revision.
LL: Corresponding author, supervision, analysis and critical review.
All authors approved the final version of the manuscript to be published.
REFERENCES
- Facchetti F, Lorenzi L. Follicular dendritic cells and related sarcoma. Semin Diagn Pathol. 2016; 33(5):262-76. doi: 10.1053/j.semdp.2016.05.002.
- Wu A, Pullarkat S. Follicular dendritic cell sarcoma. Arch Pathol Lab Med 2016; 140(2):186-90. doi: 10.5858/arpa.2014-0374-RS.
- Kim-Orden N, Chambers T, Sinha U. Rapidly enlarging parapharyngeal mass. JAMA Otolaryngol Head Neck Surg 2018; 144(12):1178-9. doi: 10.1001/jamaoto.2018.2371.
- Saygin C, Uzunaslan D, Ozguroglu M, Senocak M, Tuzuner N. Dendritic cell sarcoma: A pooled analysis including 462 cases with presentation of our case series. Crit Rev Oncol Hematol 2013; 88(2):253-71. doi: 10.1016/j.critrevonc.2013.05.006.
- Leipsic JA, McAdams HP, Sporn TA. Follicular dendritic cell sarcoma of the mediastinum. AJR Am J Roentgenol 2007; 188(6):W554-6. doi: 10.2214/AJR.04.1530.
- Long-Hua Q, Qin X, Ya-Jia G, Jian W, Xiao-Yuan F. Imaging findings of follicular dendritic cell sarcoma: report of four cases. Korean J Radiol 2011; 12(1):122-8. doi: 10.3348/kjr. 2011.12.1.122.
- Singer AD, Weinberger PM, Seybt MW, Jackson LL, Teague DJ, Figueroa RE. Imaging characterisation of follicular dendritic cell sarcoma of the neck. Ear Nose Throat J 2010; 89(7): 296-7. PMID: 20628983.
- McClelland E, Bashyam A, Derbyshire S, Di Palma S. Follicular dendritic cell sarcoma presenting as a painless lump in the parotid. BMJ Case Rep 2018; 2018. doi: 10.1136/bcr- 2018-224301.
- Chan JK, Fletcher CD, Nayler SJ, Cooper K. Follicular dendritic cell sarcoma. Clinicopathologic analysis of 17 cases suggesting a malignant potential higher than currently recognised. Cancer 1997; 79(2):294-313. PMID: 9010103.