Posterior Fascicular Ventricular Tachycardia: A Rare Cause of Tachycardia
By Erdem Kurt1, Rohat Ak2, Suphi Bahadirli3Affiliations
doi: 10.29271/jcpsp.2022.06.804ABSTRACT
A 13-year male patient with a history of tachycardia attacks was diagnosed to have left posterior fascicular ventricular tachycardia (VT) according to the electrocardiogram (ECG) obtained at the emergency service. This diagnosis was confirmed with advanced electrophysiological studies and the case was diagnosed by genetic evaluation, which was performed to reveal the underlying cause, to have Brugada type 2 syndrome that might be associated with sudden cardiac death. Underlying causes should be evaluated, although idiopathic VTs generally have a good prognosis.
Key Words: Brugada syndrome, Posterior fascicular ventricular tachycardia, Right bundle branch block.
INTRODUCTION
The underlying cause of ventricular tachycardia (VT) is generally structural heart diseases. However, 10% of patients with VT are found to have no structural heart disease, metabolic / electrolyte anomaly, or long QT syndrome and thus these arrhythmias are called idiopathic VTs. Idiopathic VT is typically a benign type of tachycardia observed in patients between 15 and 40 years of age. Symptoms include palpitations, fatigue, shortness of breath, dizziness, and presyncope. Syncope and sudden death are very rare. Most attacks occur at rest, but these can be triggered by exercise and emotional stress.1
There are three subtypes of idiopathic VTs. 1. Posterior fascicular VT (90-95% of cases): Right bundle branch block (RBBB) morphology and left axis deviation, and it occurs close to the left posterior fascicle. 2. Anterior fascicular VT (5-10% of cases): RBBB morphology and right axis deviation, and it occurs near the left anterior fascicle. 3. Upper septal fascicular VT (rare): Atypical morphology usually RBBB but may resemble left bundle branch block; narrow QRS and normal axis cases have also been reported. It comes out above the septum.1 The aim of this case report is to describe the fascicular VT subtype and to evaluate the advanced tests and treatment of this subtype.
This idiopathic VT was first described in 1979 by Zipes et al. It was characterised as a condition induced by atrial pacing and an RBBB morphology-like appearance with left axial deviation in patients without structural heart disease.2 In 1981, Belhassen et al. showed that this VT form could be terminated by verapamil and this was reported as the fourth defining feature of the syndrome.3
Fascicular VTs are often misdiagnosed as supraventricular tachycardias with a bifascicular block.4 The diagnosis and treatment approach is changed due to this misdiagnosis. To prevent this, the ECG findings of these patients should be examined carefully and advanced diagnostic methods should always be used.
CASE REPORT
A 13-year male was admitted to the emergency department with palpitations and shortness of breath. The patient gave a nonspecific medical history with no active use of any medications. He stated that he had similar complaints before and that they had resolved spontaneously. There was no history of syncope and no family history of cardiac disease.
Vital signs at presentation were as follows: Blood pressure (BP): 110/70 mmHg; heart rate: 213 / min, SO2: 97%; and temperature: 36.4°C. On cardiac examination, S1 and S2 were normal with no murmurs. Pulmonary auscultation was normal. The electrocardiogram (ECG) of the patient revealed a large complex tachycardia in a range of 120-140 ms at 213 / min pulse. A detailed examination of his ECG revealed suspected posterior fascicular VT and he was treated with intravenous diltiazem. Normal sinus rhythm was achieved in the patient whose tachyarrhythmia was terminated (Figure 1). Hemoglobin, liver function tests, renal function tests, serum electrolytes, and thyroid-stimulating hormone (TSH) were normal among the laboratory tests. Transthoracic echocardiography showed no structural heart disorders.
The patient was consulted by a pediatric cardiologist and was hospitalised for further examination and evaluation. The results of the electrophysiological study (EPS) were compatible with a left posterior fascicular VT. After genetic study, a heterozygous variation in the A208V variant of the GPD1L gene was found and the patient was diagnosed to have Brugada type 2 syndrome. The patient was successfully treated by radiofrequency ablation.
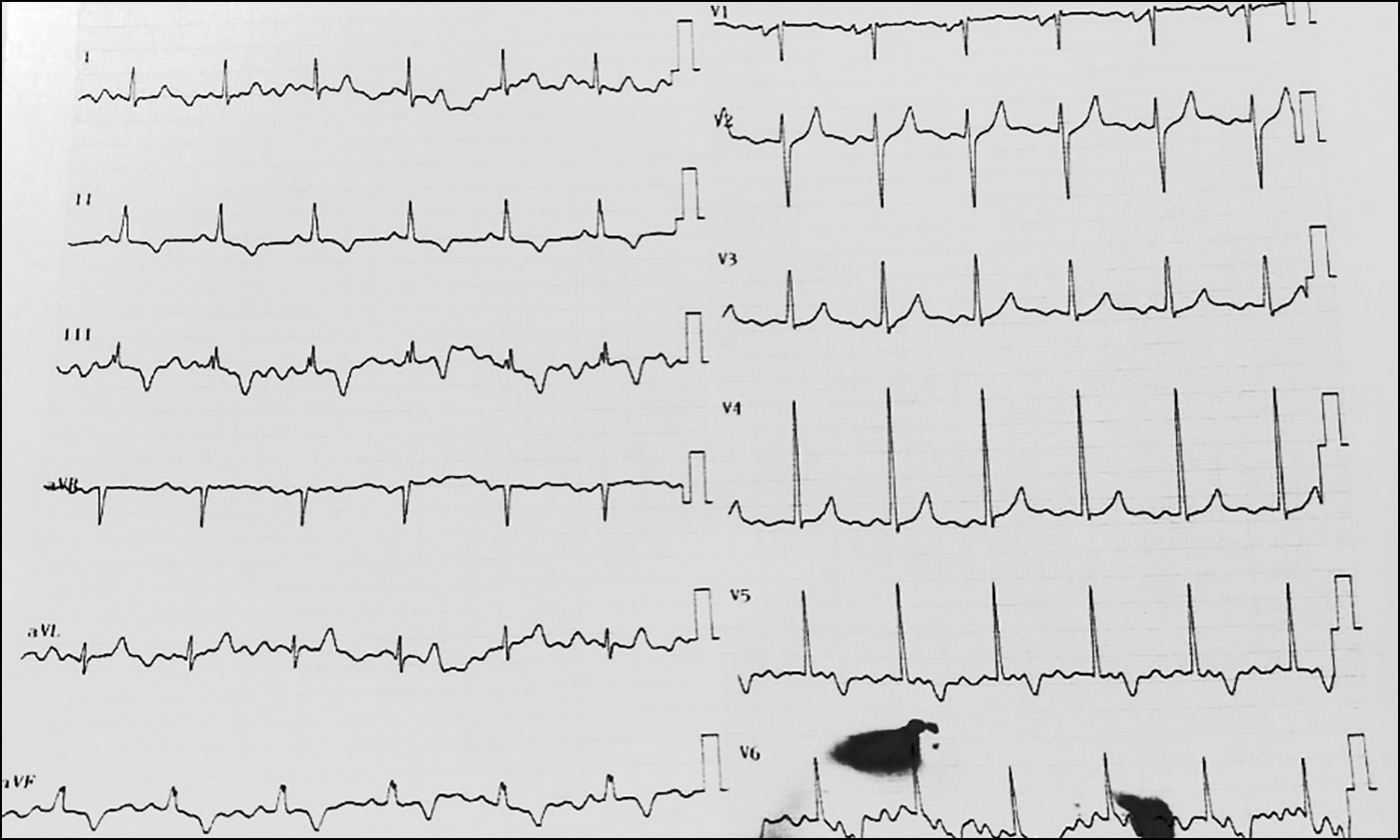 Figure 1: ECG with 215 beats per minute (bpm), absence of regular P waves, QRS complex >120 ms, RBBB-like morphology, and shift to the left of the frontal axis.
Figure 1: ECG with 215 beats per minute (bpm), absence of regular P waves, QRS complex >120 ms, RBBB-like morphology, and shift to the left of the frontal axis.
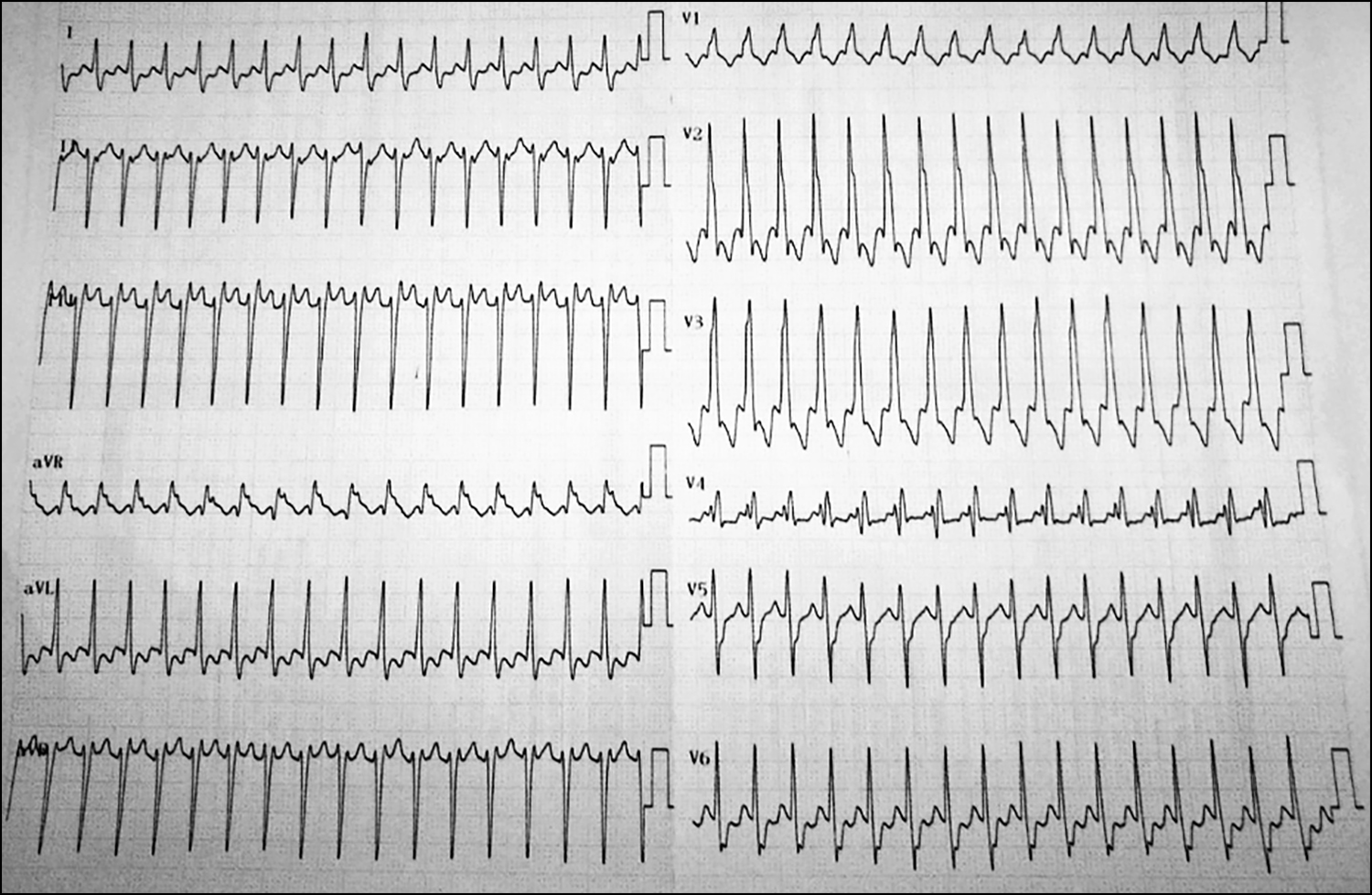 Figure 2: Atrio-ventricular (AV) dissociation is observed in lead D2, QRS complex in lead V1 and Marriott wave in lead V2.
Figure 2: Atrio-ventricular (AV) dissociation is observed in lead D2, QRS complex in lead V1 and Marriott wave in lead V2.
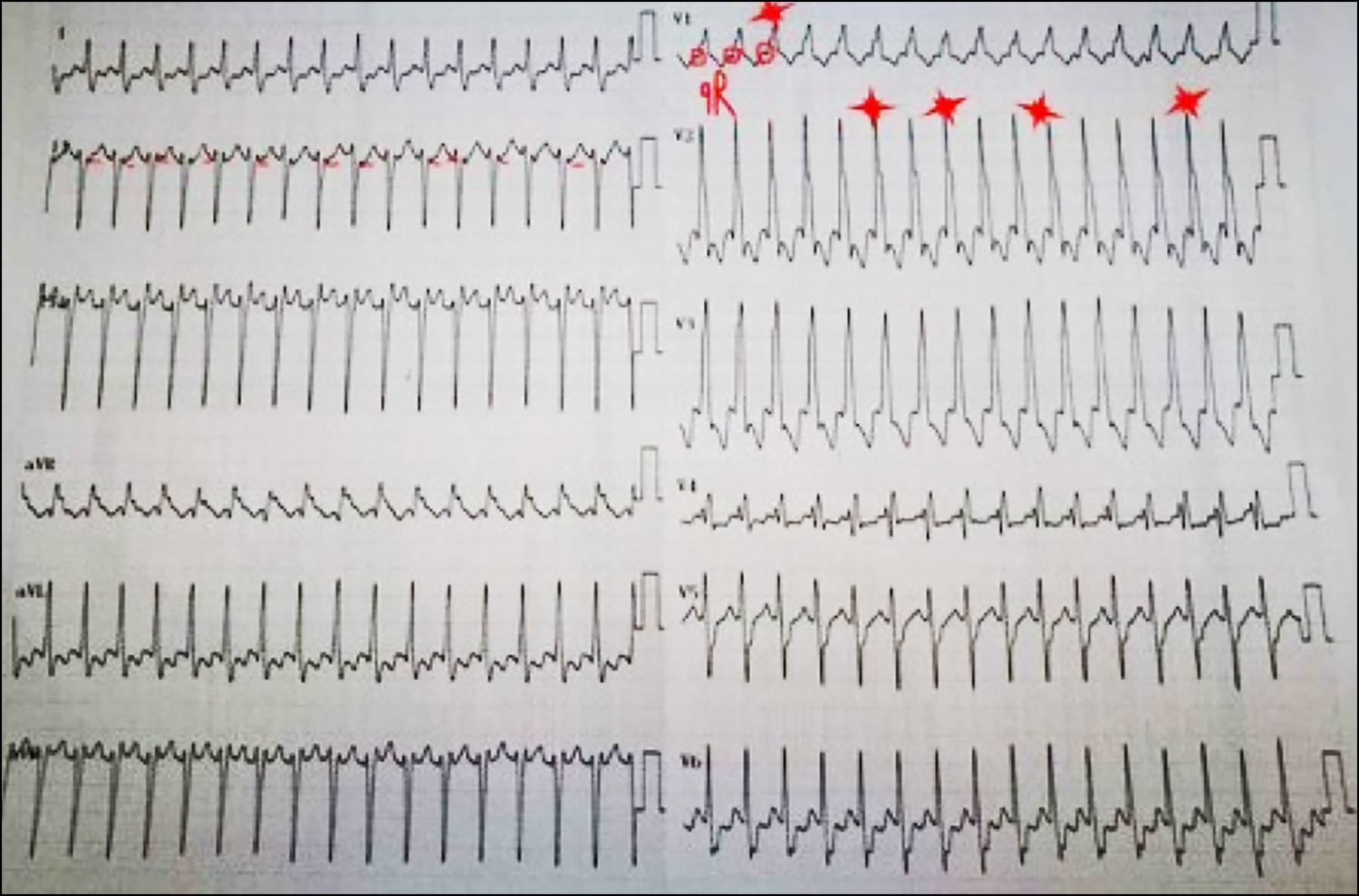 Figure 3: ECG revealed normal sinus rhythm, normal range axis after antiarrhythmic agent was administered.
Figure 3: ECG revealed normal sinus rhythm, normal range axis after antiarrhythmic agent was administered.
DISCUSSION
Fascicular tachycardia is the most common idiopathic VT of the left ventricle (LV) and is a re-entrant tachycardia due to an ectopic focus in the LV. Diagnosis can be difficult and this rhythm is often misdiagnosed as an RBBB SVT. The diagnosis is made by observing the characteristics of the VT. Fascicular tachycardia can be classified based on the ECG morphology reflecting the anatomical localisation of the re-entrant loop. Among these cases, 90-95% are posterior fascicular VTs with detection of a RBBB morphology and left axial deviation and arise from a point close to the left posterior fascicule. Another 5-10% are anterior fascicular VTs with detection of an RBBB morphology and right axial deviation and arise from a point close to the left anterior fascicule. In very rare cases, an upper septal fascicular VT is detected. It also has atypical morphology and is usually associated with RBBB; however, it may resemble the left bundle branch block (LBBB) as well. Idiopathic fascicular tachycardia is often difficult to treat when it is unresponsive to medical therapy.1
There were no regular P waves in the ECG of this case. Large QRS complexes in a range of 120-140 ms were seen in precordial derivations (Figure 2). When we see a tachycardia with a large QRS, the first question to be asked is whether this tachyarrhythmia is VT or SVT-aberrant conduction (accessory pathway, RBBB / LBBB, or functional blocking associated with rate) that causes tachycardia with wide QRS. In any case, assuming the presence of VT in all such cases is the best approach to avoid fatal results when one cannot analyze the ECG adequately. RBBB or LBBB morphology seen in aberrant conductions generally demonstrates typical branch block morphology, while it is not compatible with this typical morphology in VTs. This is a significant criterion in the differentiation of these two types of arrhythmias; however, it is better to search for the presence of other criteria as well. QRS complexes that do not resemble exactly the right bundle branch morphology can be seen easily when the ECG of the case is carefully analyzed. It is also seen that the frontal axis is -90-100 degrees.
Table I: Brugada and Vereckei algorithms.
|
Brugada Algorithm |
Vereckei Algorithm |
|
Absence of RS complexes in all precordial leads |
Initial R wave in aVR |
|
A range of RS in the precordial lead >100 ms |
Presence of the first Q or R wave >40 ms |
|
Atrioventricular (AV) dissociation |
On the descending arm of the QRS wave with negative onset presence of a notch |
|
Morphological criteria for ventricular tachycardia (VT) |
Vi / Vt<1 ( The ratio of the amplitude change in the first 30 ms and the last 30 ms of QRS) |
When the ECG is examined in more detail, the analysis of the large QRS rhythms should be based on the algorithms of differentiation of SVT with aberrant conduction and VT. The most frequently used algorithms are Brugada and Vereckei algorithms and are demonstrated in Table I.5,6
When the ECG of the present case is examined using these algorithms (Figure 3), we see that P-QRS waves are at different rates, especially in V2 and aVR, thus the presence of atrio-ventricular (AV) dissociation. In addition, the presence of a Q wave with a small QR complex and long R wave (a specific sign of VT in a right bundle block-like morphology) and the presence of the Marriott sign (appearance of the R wave as a rabbit ear with a lower notch and long left side) are highly specific for VT.7
Table II: ECG characteristics of and treatment regimens for the subtypes of idiopathic ventricular tachycardias (VTs).
|
Type of VT |
QRS morphology / Axis |
Drug therapy |
Treatment |
|
RVOT VT (75-90%) / monomorphic extrasystoles |
LBBB / inferior Axis |
Adenosine, |
RF ablation |
|
LVOT VT (5%) |
S wave in DI, R wave transition in V1-V2 |
Adenosine, |
RF ablation |
|
Fascicular VT (10-15%) |
RBBB / left axis (output posterior fascicle); RBBB / right axis (output anterior fascicle); output from upper septal location |
Verapamil |
RF ablation |
These data revealed that the tachyarrhythmia of this ECG did not resemble a typical VT. However, the wave structures of the AV dissociation and unipolar derivations not resembling the RBBB morphology and the presence of a left axis directed us to think of VT instead of SVT-aberrant conduction.
The present ECG was not typical of a frequently encountered VT, idiıoathic VT. These arrhythmias include specific ECG patterns that are helpful to define the anatomic localizations. Table II shows the ECG characteristics of and treatment regimens for the subtypes of idiopathic VTs.1
Idiopathic VTs normally have a narrower QRS waves. The distance between the start of the R wave and the lower end of the descending branch of the S wave can be as low as 60-80 ms, while it is 100 ms in other types of VTs. It was 60-80 ms in the type of VT seen in this case.6
The long-term prognosis of patients with non-structural heart disease with a fascicular VT is very good. Arrhythmias in sporadic and well-tolerated fascicular VT attacks may not progress, even though no pharmacological treatment is provided.8 Unlike other idiopathic VTs, posterior fascicular VTs are usually unresponsive to vagal maneuvers or adenosine or beta-blockers. This demonstrates that posterior fascicular VTs are due to the slow entry of calcium in partially depolarized Purkinje fibers.9
Radiofrequency catheter ablation is an appropriate treatment strategy for patients with severe symptoms or intolerance or resistance to antiarrhythmic therapy.10
Considering the etiology of idiopathic VTs, Brugada syndrome should be kept in mind. The gene analysis of the patient, in this case, revealed a heterozygous A208V variant of the GPD1L gene and the patient was diagnosed to have Brugada type 2 syndrome.
Brugada syndrome is characterised by a specific ECG pattern of the RBBB and an ST-segment elevation in early precordial leads without any structural abnormality of the heart. It causes idiopathic VT or ventricular fibrillation (VF) and carries a high risk of sudden cardiac death.11 The syndrome can be caused by many genes. At least nine genes are known to cause Brugada syndrome (SCN5A, GPD1L, CACNA1C, CACNB2, SCN1B, KCNE3, SCN3B, HCN4, and KCND3); among these, SCN5A accounts for about 20% of cases, while other "small" genes are also present comprising 15% of the cases.12
In conclusion, fascicular VT should be considered in cases with tachycardia attacks that were initially diagnosed as SVT but did not cease by standard methods and adenosine administration and present a right bundle branch-like morphology together with a left axial derivation in 12 lead ECG.
PATIENT’S CONSENT:
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
COMPETING INTEREST:
The authors declared no competing interest.
AUTHORS’ CONTRIBUTION:
EK, RA, SB: Contributed to the design and data collection, literature review, and the writing of the manuscript.
All authors approved the final version of the manuscript to be published.
REFERENCES
- Brugada J, Diez P. How to recognise and manage idiopathic ventricular tachycardia. E-J ESC Council Cardiol Prac 2010; 8.
- Zipes DP, Foster PR, Troup PJ, Pedersen DH. Atrial induction of ventricular tachycardia: Reentry versus triggered automaticity. Am J Cardiol 1979; 44(1):1-8. doi: 10.1016/ 0002-9149(79)90242-x.
- Belhassen B, Rotmensch HH, Laniado S. Response of recurrent sustained ventricular tachycardia to verapamil. Heart 1981; 46(6):679-82. doi: 10.1136/hrt.46.6.679.
- Lerman BB, Stein KM, Markowitz SM. Mechanisms of idiopathic left ventricular tachycardia. J Cardiovas Electrophysiol 1997; 8(5):571-83. doi: 10.1111/j.1540- 8167.1997.tb00826.x.
- Brugada P, Brugada J, Mont L, Smeets J, Andries EW. A new approach to the differential diagnosis of a regular tachycardia with a wide QRS complex. Circulation 1991; 83(5):1649-59. doi: 10.1161/01.cir.83.5.1649.
- Vereckei A, Duray G, Szénási G, Altemose GT, Miller JM. New algorithm using only lead aVR for differential diagnosis of wide QRS complex tachycardia. Heart rhythm 2008; 5(1):89-98. doi: 10.1016/j.hrthm.2007.09.020
- Reddy V, Kundumadam S, Kathi P, Dhillon K, Ismail H, Anem G. The exception to Marriot's sign. Ann Noninvasive Electrocardiol 2017; 22(5):e12449. doi: 10.1111/anec. 12449.
- Ohe T, Aihara N, Kamakura S, Kurita T, Shimizu W, Shimomura K. Long-term outcome of verapamil-sensitive sustained left ventricular tachycardia in patients without structural heart disease. J Am Coll Cardiol 1995; 25(1): 54-8. doi: 10.1016/0735-1097(94)00324-j.
- Lerman BB. Response of nonreentrant catecholamine-mediated ventricular tachycardia to endogenous adenosine and acetylcholine. Evidence for myocardial receptor-mediated effects. Circulation 1993; 87(2):382-90. doi: 10. 1161/01.cir.87.2.382.
- Nakagawa H, Beckman KJ, McClelland JH, Wang X, Arruda M, Santoro I, et al. Radiofrequency catheter ablation of idiopathic left ventricular tachycardia guided by a Purkinje potential. Circulation 1993; 88(6):2607-17. doi: 10.1161/ 01.cir.88.6.2607.
- Letsas KP, Weber R, Astheimer K, Kalusche D, Arentz T. T peak–T end interval and T peak–T end/QT ratio as markers of ventricular tachycardia inducibility in subjects with Brugada ECG phenotype. Europace 2009; 12(2):271-4. doi: 10.1093/europace/eup357.
- Mademont-Soler I, Riuró H, Mates J, Pérez-Serra A, Coll M, Porres JM, et al.Large genomic imbalances in Brugada syndrome. Plos One 2016; 11(9):e0163514. doi:10.1371/ journal.pone.0163514.