Papillary Urothelial Carcinoma Arising from Ureterocele: Is it a Rare Occurrence?
By Ayhan Karakose1, Ekrem Akdeniz2Affiliations
doi: 10.29271/jcpsp.2022.10.1360ABSTRACT
Various hydronephrotic, dysplastic or clinical pathologies can accompany ureterocele, which is a cystic dilatation of the intravesical part of the submucosal ureter. However, the development of cancer in the ureterocele is highly unusual. Hematuria is the most common sign, although imaging can also indicate alterations in the wall of the ureterocele. Unfortunately, there is no current guideline for the management of papillary urothelial carcinoma arising from ureterocele. Non-muscle-invasive bladder cancer treatment and follow-up protocols are classically applied. However, the anatomical structure of the ureterocele differs from that of normal bladder tissue. It is unclear whether this difference represents a risk in terms of deep tissue tumour invasion. In addition, tumour cells may migrate to the upper urinary tract due to urinary reflux developing following tumour resection. The management of papillary urothelial carcinoma arising from ureterocele must therefore be specific to that condition. However, this depends on an increase in the number of patients reported in the literature. Our review of the literature revealed very few reports. The present study is therefore particularly valuable from that perspective and describes the clinical management of a patient developing papillary urothelial carcinoma in ureterocele.
Key Words: Ureterocele, Bladder, Hematuria, Urothelial carcinoma.
INTRODUCTION
A ureterocele is a congenital abnormality representing a pseudo-cystic dilatation of the last segment of the ureter in the bladder wall.1,2 It is usually detected when investigating patients with lower urinary tract symptoms (LUTS) or infections.3,4
Urothelial cancer is most often detected in the bladder, and hematuria is the most common presenting feature. It is very rare and unusual for ureterocele to be associated with tumour. However, there are a few reported cases of malignant tumours of ureterocele, particularly urothelial carcinomas.1
We report a rare case of papillary urothelial carcinoma arising from ureterocele. Bipolar transurethral resection (TUR) was performed without complications. Postoperative intravesical immunotherapy was initiated with Bacillus Calmette-Guérin (BCG) in six weekly sessions.
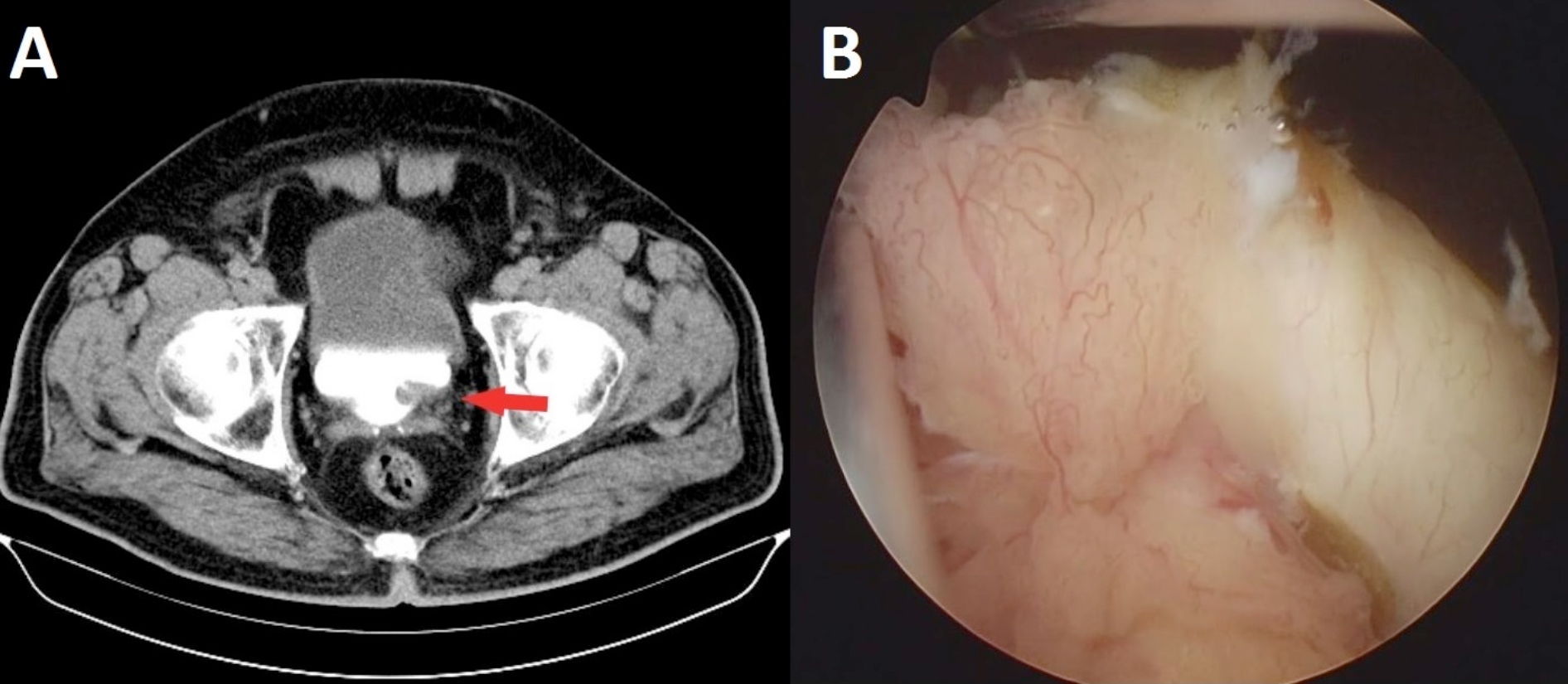 Figure 1: Radiological and endoscopic view of the tumour (A, Preoperative excretory phases of the CT urography image with ureterocele containing soft tumoural tissue [red arrow]; B, Solid papillary lesion on the ureterocele detected at cystoscopy).
Figure 1: Radiological and endoscopic view of the tumour (A, Preoperative excretory phases of the CT urography image with ureterocele containing soft tumoural tissue [red arrow]; B, Solid papillary lesion on the ureterocele detected at cystoscopy).
CASE REPORT
A 65-year male patient presented to the urology outpatient clinic with intermittent macroscopic hematuria and suprapubic pain persisting for one month. Urine cytology, blood values, and serum prostate specific antigen (PSA) were normal. Physical examination was also normal. Computed tomography (CT) urography revealed a simple ureterocele containing soft tumoural tissue. Cystoscopy revealed a papillary lesion on the anterior wall of the ureterocele (Figure 1). No other lesions were observed elsewhere in the bladder.
The patient read and filled the comprehensive approved written informed consent which was appropriately declaring surgical procedures, possible risks, and benefits in preoperatively. Bipolar TUR was performed without complications. Histopathological diagnosis revealed non-invasive low-grade papillary urothelial carcinoma (Figure 2). Postoperative intravesical immunotherapy was initiated with BCG in six weekly sessions (40 mg). Maintenance of BCG in three weekly sessions (40 mg) was planned for three years. To date, the patient has been followed-up with periodic serum kidney function tests, urine cytology, imaging, and urethrocystoscopic studies with no recurrence or progression of the disease.
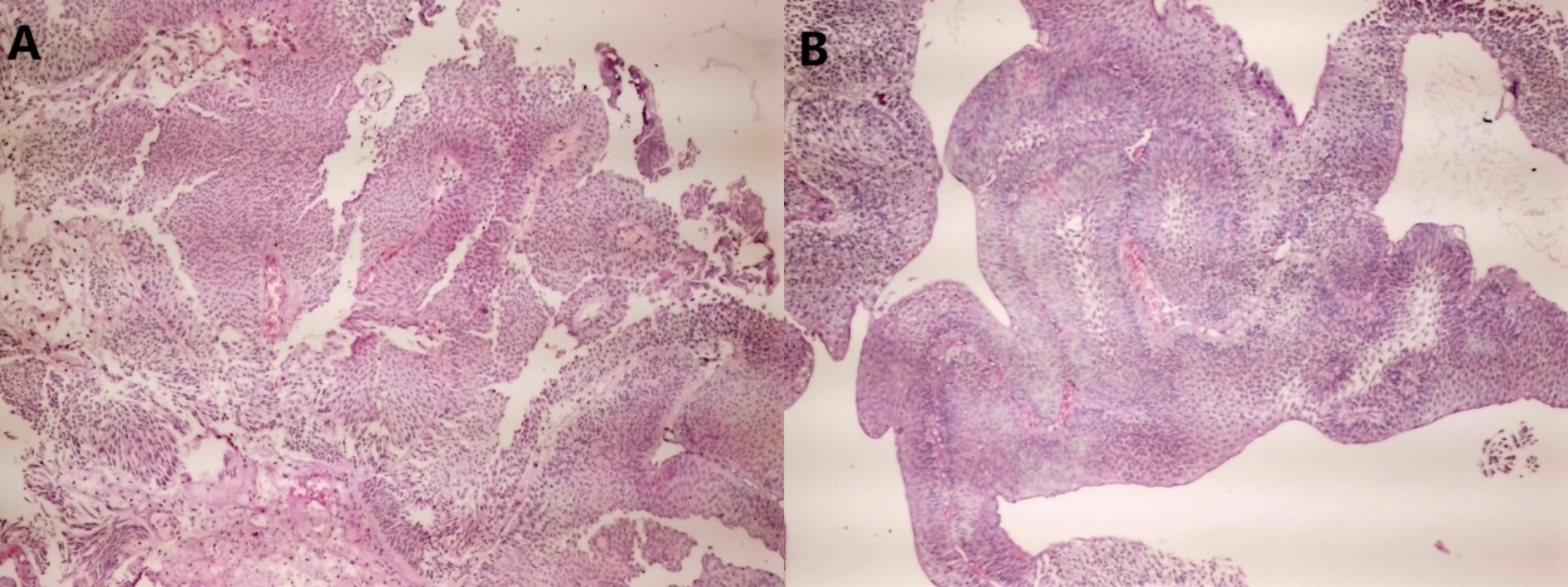 Figure 2: Pathological findings from the tumour (A, non-invasive low-grade papillary urothelial carcinoma area [HE; ×40]; B, papillary tumour consisting of cells with mild cytological atypia and disordered arrangement around the fibrovascular core [HE; ×40]).
Figure 2: Pathological findings from the tumour (A, non-invasive low-grade papillary urothelial carcinoma area [HE; ×40]; B, papillary tumour consisting of cells with mild cytological atypia and disordered arrangement around the fibrovascular core [HE; ×40]).
DISCUSSION
Although ureterocele, representing cystic dilatation of the intravesical part of the submucosal ureter, is generally asymptomatic it may also be accompanied by various diseases, such as infection, pelvicalyceal ectasia, kidney disease, and by congenital pathologies including vesicoureteral reflux, ureteropelvic duplication, and renal dysplasia. The aetiology of ureterocele is unclear, but the entity is seen in an average one in 4000 births.5 Although, it can be diagnosed in the intrauterine period due to widespread ultrasound use, ureterocele is generally diagnosed in adulthood, and orthotopic ureterocele is usually accompanied by LUTS.5 Imaging studies are the most effective method of diagnosing ureterocele and lesions contained inside it.
Ureterocele with accompanying papillary urothelial carcinoma is a highly unusual entity, and to the best of our knowledge, this is limited to a few case reports.6 The most common symptoms in previous reports are LUTS and hematuria.1,2 Tumours derived from the ureterocele exhibiting various characteristics of irregular echogenicity with no acoustic shadowing may be observed at sonographic examination.7 At CT urography, ureterocele is typically characterised by an intravesical defect with a radiolucent and globular appearance in the form of the “cobra-head sign.”1 This modality is particularly useful for visualising enhancing masses such as tumours, as well as permitting the exclusion of extravesical disease. Cystoscopic examination must be conducted without loss of time if the appearance is not ‘classic.’ This patient’s principal symptom was LUTS + hematuria. Ureterocele together with interior soft tissue mass lesion was observed on the CT urogram, and the diagnosis was confirmed with cystoscopy.
Nephroureterectomy, resection of the ureterocele with a segment of the terminal ureter and re-implantation, intracavitary chemotherapy, or simple observation and monitoring have all been described for definitive treatment.1,6 There is no current guideline available for the management of papillary urothelial carcinoma arising from ureterocele. Non-muscle-invasive bladder cancer (NMIBC) treatment and follow-up protocols are classically applied. In the present case, the ureterocele was unroofed for the purpose of visual confirmation, followed by resection for clearance. The pathology report indicated low-grade urothelial carcinoma with no observation of suburothelial or muscular compromise. The patient then received BCG therapy. We planned to administer BCG maintenance treatment for three years, with periodic urinary cytology, imaging, and urethrocystoscopic studies.
Histological examination may reveal varying amounts of attenuated smooth muscle bundles and fibrous tissue in the ureterocele wall. Externally, the ureterocele is surrounded by vesical mucosa, while being lined internally with ureteric mucosa.8 It is unclear whether this will constitute an advantage or a disadvantage in terms of deep tissue tumour invasion. In addition, it may cause reflux of tumour cells developing after unroofing to migrate to the lower urinary tract. The standard NMIBC follow-up protocol may not be suitable for these patients. Urethrocystoscopy alone may not be sufficient, and cystoureterorenoscopy may be more appropriate. In addition, imaging techniques such as CT urography and sonography may need to be repeated at more frequent intervals. These factors represent an area of uncertainty in the management of the disease and are problematic for clinicians. Papillary urothelial carcinoma arising from ureterocele should be managed specifically and separately from NMIBC, although this depends on an increase in the number of patients reported in the literature. However, an appropriate treatment/follow-up chart can be produced once a sufficient number of patients has been achieved.
CONCLUSION
Urothelial carcinoma development in ureterocele is exceedingly unusual and no current guidelines are available for its management. Transurethral resection facilitates pathological diagnosis and a definite treatment decision. Due to its distinctive histological structure, an appropriate management protocol for papillary urothelial carcinoma arising from ureterocele needs to be determined.
PATIENT’S CONSENT:
Informed consent was obtained from the patient.
COMPETING INTEREST:
The authors declared no competing interest.
AUTHORS’ CONTRIBUTION:
AK: Establishment and confirmation of the diagnosis, analysis of data for the manuscript, drafting and revising the intellectual content of the manuscript, final approval, and agreement to be accountable for all aspects of the manuscript.
EA: Design of the manuscript, literature search, drafting and revising the intellectual content of the manuscript, final approval, and agreement to be accountable for all aspects of the manuscript.
REFERENCES
- Astigueta JC, Abad-Licham M, Silva E, Alvarez V, Piccone F, Cruz E, et al. Ureterocele urothelial carcinoma: managing a rare presentation. Ecancer medical science 2016; 10:621. doi: 10.3332/ecancer.2016.621.
- Xie D, Klopukh B, Nehrenz GM, Gheiler E. Ureterocele: Review of Presentations, Types and Coexisting Diseases. Int Arch Urol Complic 2017; 3:024. doi: 10.23937/2469- 5742/1510024.
- Atesci YZ, Aydogdu O, Karakose A, Pekedis M, Karal O, Senturk U. Does urinary bladder shape affect urinary flow rate in men with lower urinary tract symptoms? Scientific World J 2014; 2014:846856. doi: 10.1155/2014/846856.
- Karakose A, Alp T, Güner ND, Çıtlak MB, Aydın A. The place of andrological cases in our general urology practice. Turk J Urol 2010; 36(1):49-54. doi: 10.1155/2014/846856.
- Paediatric urology. European Association of Urology. uroweb.org/guideline/paediatric-urology/#3_15. (Accessed on 25/07/2020).
- Law YXT, Sagir IK, Tan GLL. A solitary urothelial tumour arising from one of bilateral ureteroceles. Int Braz J Urol 2017; 43(5):990-3. doi: 10.1590/S1677-5538.
- Andrew WK, Thomas RG, Aitken FG. Simple ureteroceles-ultrasonographic recognition and diagnosis of compli-cations. S Afr Med J 1985; 67(1):20-2.
- Schlussel RN, Retik AB. Anomalies of the ureter. In: Walsh PC, Retik AB, Stamey TA, Vaughan ED, Eds, Campbell’s Urology. ed. 7th, Philadelphia; WB Saunders; 1998: p.1814–57.