Management of Complicated Acute Pancreatitis: The Role of Hub-and-Spoke Model
By Khalid Munir Bhatti, Shafiq Rehman, Salman Ejaz Ahmed, Ruben CaneloAffiliations
doi: 10.29271/jcpsp.2022.05.575ABSTRACT
Objective: To determine the primary and secondary outcomes of patients with complicated acute pancreatitis (CAP) of moderate to severe intensity managed by using the hub-and-spoke model.
Study Design: An observational study.
Place and Duration of Study: Department of Surgery, North Cumbria Integrated Care, Carlisle, UK, from January 2014 to December 2018.
Methodology: Retrospective analysis of 496 episodes of acute pancreatitis managed in 405 patients was done. Data for demographic features and clinical outcomes were analysed. In patients with recurrent admissions, only index admission was considered for analysis. Complicated acute pancreatitis was defined by using the revised Atlanta classification and included all the acute pancreatitis patients with local and or systemic complications.
Results: The frequency of CAP was 21.7% (88/405). The mean patients’ age was 62.11 ± 17.90 years. The intensive therapy unit (ITU) admission rate was 33% (n = 29), whereas the overall intervention rate was 43.2% (n = 38). The in-hospital mortality rate was 10.2% (n = 9), and the overall mortality rate was 14.8% (n = 13). A comparative analysis of clinical outcomes according to the revised Atlanta classification showed that the rate of complications, need for ITU admission, duration of hospital stay, in-hospital mortality and overall mortality were significantly higher in patients with moderately severe AP (MSAP) and severe AP (SAP).
Conclusion: The rate of progression from mild AP to MSAP and SAP remains high. Patients with CAP are at higher risk of ITU admission, prolonged hospital stay, in-hospital mortality and overall mortality. To improve clinical outcomes, the progression of AP to severer forms should be prevented by developing newer strategies, and in cases where complications have already developed, the mortality rate needs to be improved by developing innovative treatment modalities.
Key Words: Acute pancreatitis, Complicated acute pancreatitis, Revised Atlanta classification, Morbidity, Mortality, Survival analysis, Hub and spoke model.
INTRODUCTION
Acute pancreatitis (AP) is a common surgical presentation with a reported incidence of 13–45 per 100,000 people, annually.1 Progression of AP to moderately severe AP (MSAP) and severe AP (SAP) is of major concern owing to the higher morbidity and mortality associated with MSAP and SAP.2,3 This increased risk of morbidity and mortality has been attributed to inconsistencies in identifying and classifying the severity of AP, resulting in a lack of a standard of care.1,4
The revised Atlanta classification of acute pancreatitis (2012) was an update of the 1992 Atlanta classification.2 The revised guideline was aimed at achieving international consensus regarding the severity of pancreatitis and its complications; and both local and systemic complications formed the basis of this classification. Mild AP (MAP) is not associated with local or systemic complications; however, MSAP is defined as the presence of local complications or transient organ failure of less than 48 hours.2 Acute pancreatitis in the presence of persistent organ failure of more than 48 hours defines SAP.2 MSAP and SAP can be classified together as complicated acute pancreatitis (CAP) because these forms involve complications; i.e., local and/or systemic.5
Managing CAP is challenging as it is associated with a prolonged hospital stay and significant morbidity and mortality of up to 40%.5 To improve outcomes, the International Association of Pancreatology (IAP) has suggested a hub-and-spoke model for the management of patients with CAP.1 According to the guidelines, all patients presenting at local hospitals (spokes) with CAP should receive regular input from specialist centres (hub).1,6 The decision regarding transfer of these patients should be pragmatic and directed by two factors: the type of complications and the capability of the primary hospital to handle these complications.1,6 This hub-and-spoke model for the management of CAP appears to be resource-efficient, as most patients will not require transfer to specialist centres. However, the clinical outcomes of patients with CAP managed at primary hospitals are not frequently reported.7,8 Reporting outcomes such as complications, need for intensive therapy unit (ITU) admission, length of hospital stay, intervention rate, in-hospital mortality and long-term mortality will highlight the adequacy of the management in primary hospitals.
The current study aimed to report the clinical outcomes of patients with CAP managed at a local hospital using the hub-and-spoke model over a 5-year period.
METHODOLOGY
This observational study involved a retrospective analysis of data submitted to a local database for all patients with AP admitted at North Cumbria Integrated Care, Carlisle, UK, between January 2014 and December 2018. The inclusion criterion was the diagnosis of AP as described in the IAP Working Group and American Pancreatic Association (APA) guidelines.1 CAP was defined according to the revised Atlanta classification by the presence of any local or systemic complications.2 Where the patients had multiple admissions, only index admission was included for final analysis. The primary outcomes were in-hospital mortality, and the secondary outcomes were the need for ITU admission, the need for radiological or surgical interventions, and the development of chronic pancreatitis and mortality at the end of the study period. Hospital records were used, to determine the mortality.
Other variables were the year of admission, age, gender, comorbidities, findings on computed tomography (CT) and the need for endoscopic retrograde cholangiopancreatography (ERCP). Ward-based patient care involving intravenous (IV) fluids, analgesia and nutritional support was defined as conservative management, while symptomatic treatment with an end-of-life care pathway was defined as supportive care. End-of-life care decisions were made by senior clinicians in consultation with intensivists, according to various parameters, such as the patient’s current clinical condition, disease severity, comorbidities, functional status, presence of organ failure and the futility of ITU support. These decisions also involved honest discussions with the patient’s relatives and/or with the patients.
The care of all patients with CAP was discussed regularly with specialist centres. The criteria for referral to a specialist centre included moderately severe and severe acute pancreatitis on revised Atlanta classification, irrespective of the need for physical transfer. As per guidance, such patients were managed by the specialist centre remotely by reviewing their biochemical profile and imaging and providing input to spoke hospital on weekly basis. This process continued until physical transfer was required for interventions exceeding the resources of spoke hospital or the patient is discharged after recovery. Only the patient requiring endoscopic, video-assisted, laparoscopic or open management of pancreatic pseudocyst or walled-off necrosis, were physically transferred to a specialist centre.
Data were analysed using the Statistical Package for the Social Sciences (SPSS), Version 20 (IBM Corp., Armonk, NY, USA). Descriptive statistics were used to analyse the different variables. Means with standard deviations (SD) and median (minimum-maximum) were used for numerical variables. Frequencies and percentages were determined for categorical variables and comparisons were done by chi-square or likelihood ratio test. Kaplan–Meier survival analysis was used to determine the percentage survival during the follow-up. A p-value of <0.05 was considered significant.
RESULTS
A total of 496 episodes of AP were managed in 405 patients over the 5-year period of the study. The frequency of CAP was 21.7% (88/405). The number of patients with MSAP was 58 (65.9%), and 30 (34.1%) had SAP. The age of the patients with CAP ranged from 21 to 97 years with a mean of 62.11 ± 17.90 years. The majority of the patients (62.5%, n = 55) were more than 60 years of age, and the male-to-female ratio was 1:1. An underlying aetiology was identified in 67 cases (76.1%), while the remaining patients (n = 21, 23. 9%) had idiopathic pancreatitis. The commonest cause of CAP was biliary acute pancreatitis (n = 47; 53.4%). Other types included alcohol-induced pancreatitis (n = 20, 22.7%), and acute pancreatitis unspecified/ idiopathic (n = 21, 23.8%). Underlying comorbidities were found in 27.3% of the patients (n = 24), with the commonest comorbidity being diabetes mellitus (DM) alone or in combination with other comorbidities, such as hypertension or cardiovascular disease (13.6%, n = 12).
CT of the chest, abdomen and pelvis was performed in 74 cases (84.1%). No radiological evidence of pancreatitis was found in 1 patient although combination of symptoms and serum amylase level was very much suggestive of acute pancreatitis. The findings on CT scan in rest of the patients included acute necrotic collection (n=19, 21.6%), pseudocyst formation (n=15, 17%), acute fluid collections (n=10, 11.4%), acute pancreatitis (n=9, 10.2%), combination of acute fluid collections and acute necrotic collection (n=6, 6.8%), acute necrotic collection with pleural effusion (n=4, 4.5%), acute necrotic collection with splenic vein thrombosis (n=3, 3.4%), walled-off necrosis and pseudocyst formation (n=2, 2.3%), acute necrotic collection with portal vein thrombosis (n=3, 3.4%), acute pancreatitis with viscus perforation (n=1, 1.1%), and walled-off necrosis (n=1, 1.1%).
The management of patients with CAP is shown in Table I; approaches varied from conservative management to ITU-based organ support. Possible referral to the ITU was provided for all patients. However, 4 patients (4.5%) were determined unfit for ITU support owing to poor outcomes and entered an end-of-life care pathway; these patients died during their stay in the hospital. Three patients with MSAP and 26 patients with SAP were admitted to the ITU. One patient was admitted to the coronary care unit (CCU) owing to a cardiac event during his stay in the hospital, and he died of myocardial infarction (MI) during the same admission. Among the 29 patients admitted to the ITU, 23 (79.3%) fully recovered. Length of ITU stays varied from 2 to 35 days with a median of 7 days while hospital stay varied from 1 to 57 days with a median of 11 days. The shortest stay of one-day stay was for a patient who died within 24 hours of the presentation.
Table I: Management approaches.
|
Management |
Number of cases |
Percentage (%) |
|
Conservative management |
29 |
33 |
|
ITU management + ERCP (in- patient) + Interval cholecystectomy |
13 |
14.8 |
|
ITU management alone |
10 |
11.4 |
|
Antibiotics |
6 |
6.8 |
|
Antibiotics + Interval cholecystectomy |
4 |
4.5 |
|
End-of-life care |
4 |
4.5 |
|
CT-guided drainage |
3 |
3.4 |
|
ERCP (in-patient) |
3 |
3.4 |
|
US-guided drainage |
2 |
2.3 |
|
CT-guided drainage + Interval cholecystectomy |
2 |
2.3 |
|
Cholecystectomy on index admission |
2 |
2.3 |
|
ITU Management + ERCP (in- patient) |
2 |
2.3 |
|
ITU management + Interval cholecystectomy |
2 |
2.3 |
|
Interval cholecystectomy |
2 |
2.3 |
|
Cholecystostomy |
1 |
1.1 |
|
ITU management + Cholecystostomy |
1 |
1.1 |
|
CCU management for MI |
1 |
1.1 |
|
ERCP (out-patient) |
1 |
1.1 |
|
Total |
88 |
100 |
Antibiotics were not used in most of the patients (Table I). The indications for antibiotic therapy in CAP were guided by the NICE guidelines and the advice of the specialist centre; i.e., the hub, on a case-by-case basis. These indications were an obstructed biliary system with evidence of ascending cholangitis or positive blood cultures in an unobstructed biliary system. In most of the instances, antibiotics were started empirically and then changed to narrow-spectrum antibiotics after the results of the blood culture. Infected pancreatic necrosis was differentiated from the sterile pancreatic necrosis by the presence of air in the walled-off necrosis on cross-sectional imaging. Radiological aspiration of the necrosis for pus or tissue culture is not routinely practiced in our centre. Regarding interventional ERCP, it was not required in most of the patients because, in most of the biliary pancreatitis patients, magnetic resonance cholangiopancreatography (MRCP) showed no evidence of obstructive biliary disease.
The overall intervention rate (radiological, endoscopic and surgical) was 43.2% (n = 38). Local complications beyond 4 weeks were noted in 18 patients (20. 5%), and 10 patients (11.4 %) required transfer to a tertiary care hospital (hub) for further management. All patients referred to a tertiary centre were managed with radiological/endoscopic interventions without the need for open or laparoscopic necrosectomy. These indications included symptomatic pancreatic pseudocyst and infected or symptomatic walled-off pancreatic necrosis.
In-hospital mortality was 10.2% (n = 9) while overall mortality over the study period was 14.8 % (n= 13). Overall mortality was all-cause, and it was difficult to ascertain the cause of death in four patients who died out of our hospital during the study period. Their death was confirmed from GP records accessible through the hospital database. Duration of follow-up varied from 0 to 48 months. At mean and median follow-ups of 12 and 20 months, respectively, 85.2% of the CAP patients (n = 75) were alive. Among these, 35 (46.7%) patients developed recurrent or chronic pancreatitis. The aetiologies in recurrent/chronic cases were alcohol-induced pancreatitis (28%, n = 21), idiopathic/non-specific (13.3%, n = 10) and biliary pancreatitis (5.3%, n = 4).
Comparative analysis of clinical outcomes of patients according to the revised Atlanta classification is shown in Table II. The complications rate, need for ITU admission, duration of hospital stay, in-hospital mortality, and overall mortality were significantly higher in patients with MSAP and SAP. However, the patients with mild acute pancreatitis had a higher rate of recurrent or chronic pancreatitis (MAP = 58.5%; MSAP = 56.5%; SAP = 20%). Figure 1 shows the Kaplan–Meier survival curve for patients with AP.
DISCUSSION
The current study evaluated patients with CAP managed at a DGH, using a hub-and-spoke care model. Although this was a retrospective study, it has many strengths. First, this study evaluated both the local and systemic complications of CAP.1,2 This approach is more pragmatic because, according to the revised Atlanta classification, the presence or absence of local or systemic complications helps in clinical decision-making. Second, although there are publications reporting the management of complicated pancreatitis at a local hospital, the outcomes of this management model have not been reported frequently.7,8 Additionally, this was a single-centre study reporting a large number of patients managed for CAP documenting internal validation of the practices, and inconsistencies or variations in patient management were minimal, if any. The important findings of the current study are that we identified a high proportion of patients with progressive disease, and high morbidity and mortality in patients with AP according to disease severity.
In the current series, almost one-fifth of the patients had severer forms of the disease. Similar findings have been reported by Koutroumpakis et al. in a recent prospective study.9 According to the study, the rate for MSAP and SAP was 23.5% and 24.5% respectively, suggesting that a significant number of patients still develop the complicated disease.9-11 This progression from milder to severer forms of AP has many implications in terms of clinical, humanistic and economic outcomes.3,12,13 Regarding clinical outcomes, morbidity and mortality increases with increasing disease severity.14
|
Table II: Comparative analysis of different clinical outcomes by severity of Acute pancreatitis (n=405).
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
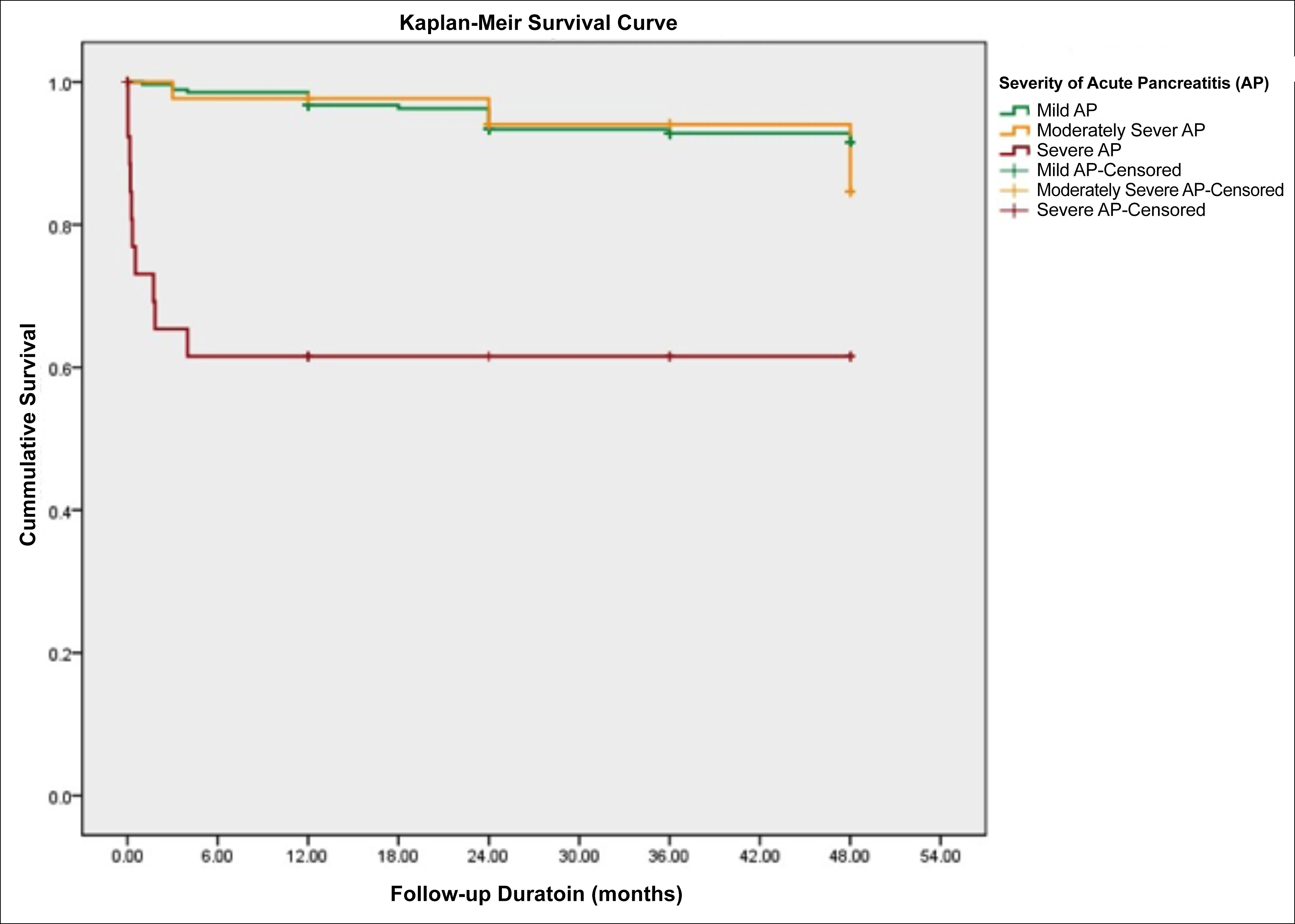 Figure 1: Kaplan-Meier curve comparing survival of patients with acute pancreatitis based on revised Atlanta classification.
Figure 1: Kaplan-Meier curve comparing survival of patients with acute pancreatitis based on revised Atlanta classification.
AP = Acute pancreatitis; MAP = Mild acute pancreatitis; MSAP = Moderately Severe acute pancreatitis; SAP = Severe acute pancreatitis
In situations where AP has already progressed to CAP, attempts should be made to prevent mortality. In 2010, an escalating step-up approach was advocated by the Dutch Pancreatitis Research Group to manage patients with CAP, aiming to reduce mortality associated with open necrosectomy in the acute phase.15 Subsequently, the same group reported the results of the pancreatitis endoscopic transgastric vs. primary necrosectomy in patients with infected pancreatic necrosis (PENGUIN) trial and the transluminal endoscopic step-up approach versus minimally invasive surgical step-up approach in patients with infected pancreatic necrosis (TENSION) trial.16,17 Although the authors reported lower morbidity, none of the above three trials reported significantly lower mortality. This situation is quite alarming as to date there are no effective measures to prevent disease progression, and similarly, there are no effective measures to reduce mortality in patients who have already developed CAP.18
To address these challenging issues, Vivian et al. recently reported 40 quality indicators to reduce morbidity and mortality, developed by the Acute Pancreatitis Task Force on Quality.19 Other strategies to prevent AP progression or to manage CAP in cases of the progressive disease are under investigation. Hines et al. in a recent review, reported new emerging trials in this regard.20 On-going trials aimed at preventing progression are the randomised treatment of acute pancreatitis with infliximab: double-blind multicentre trial (RAPID-1) and a registered trial: Clinicaltrials.gov NCT03807856. These trials will report the role of infliximab and dabigatran, respectively, in preventing AP progression. Furthermore, the early percutaneous catheter drainage of sterile pancreatic fluid collections in severe acute pancreatitis (EPCDSAP) trial is designed to assess the role of early intervention in CAP. These trials are expected to answer very important questions related to AP management.
The current study is a single centre experience and involves retrospective analysis of the data. The focus of the current study was only to report clinical outcomes. Other related issues like the financial benefits of treating the patient at the spoke hospital and psychological gains to the patients and their families due to staying close to the home, have not been considered. Similarly, advantages from avoiding the treatment of all the patients at the hub hospital should be studied through prospective designs.
CONCLUSION
Progression from MAP to MSAP and SAP remains high. Patients with MSAP and SAP are a special group of patients owing to their higher risk of ITU admission, prolonged hospital stay, in-hospital mortality and overall mortality. To improve clinical outcomes in patients with AP, newer and more effective preventive and treatment strategies need to be developed.
ETHICAL APPROVAL:
As it was non-interventional retrospective study, ethical approval was not required. However, it was registered with and approved by the Directorate for Clinical Effectiveness of North Cumbria Integrated Care (NCIC) NHS Foundation as a quality improvement project (Reg. No. 727).
PATIENT’S CONSENT:
AS the data was anonymized, individual patient’s consent was not required for the publication of the data.
COMPETING INTEREST:
The authors declared no conflict of interest.
AUTHORS’ CONTRIBUTION:
RC: Conceived the idea of the research work.
SR, SEA collected the data.
KMB: Analysed and interpreted the data; drafted and revised the content; and submitted for publication.
All authors approved the final version of the manuscript to be published.
REFERENCES
- Working Group IAP/APA acute pancreatitis guidelines. IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology 2013; 13(4):e1-15. doi: 10. 1016/j.pan.2013.07.063.
- Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, et al. Classification of acute pancreatitis - 2012: Revision of the Atlanta classification and definitions by international consensus. Gut 2013; 62(1):102-11. doi: 10.1136/gutjnl-2012-302779.
- Sarri G, Guo Y, Iheanacho I, Puelles J. Moderately severe and severe acute pancreatitis: A systematic review of the outcomes in the USA and European Union-5. BMJ Open Gastroenterol 2019; 6(1):e000248. doi: 10.1136/bmjgast- 2018-000248.
- National confidential enquiry into patient outcome and death (NCEPOD). Treat the cause: A review of the quality of care provided to patients treated for acute pancreatitis 2016 [cited 2020 Aug 18]. Available from: https://www. ncepod.org.uk/2016ap.html.
- Dickson EJ, Carter CR, McKay CJ. Complicated acute pancreatitis. In: Parks RW, Ed. Hepatobilliary and Pancreatic Surgery-A companion to specialist surgical practice. ed.6th, Edinburgh; Elsevier; 2019: p. 244-56.
- Samarasekera E, Mahammed S, Carlisle S, Charnley R. Pancreatitis: Summary of NICE guidance. BMJ 2018; 362:1-7.
- Lahiri RP, Karanjia ND. Acute necrotising pancreatitis – early management in the district general hospital and tertiary hepato-pancreatico-biliary unit. J Intensive Care Soc 2019; 20:263-7.
- Crowther N, Kahvo M, Chana P. Improving the management of acute pancreatitis in a district general hospital. BMJ Qual Improv Rep 2015; 4(1):u206577.w2642. doi: 10.1136/bmjquality.u206577.w2642.
- Koutroumpakis E, Slivka A, Furlan A, Dasyam Ak, Dudekula A, Greer JB, et al. Management and outcomes of acute pancreatitis patients over the last decade: A US tertiary-center experience. Pancreatology 2017; 17(1):32-40. doi: 10.1016/j.pan.2016.10.011.
- Harrison DA, D'Amico G, Singer M. Case mix, outcome, and activity for admissions to UK critical care units with severe acute pancreatitis: A secondary analysis of the ICNARC case mix programme database. Crit Care 2007; 1(1):S1. doi: 10.1186/cc5682.
- De Rai P, Zerbi A, Castoldi L, Bassi C, Frulloni L, Uomo G, et al. Surgical management of acute pancreatitis in Italy: Lessons from a prospective multicentre study [published correction appears in HPB 2015; 17:1049]. HPB 2010; 12:597-604.
- Soran A, Chelluri L, Lee KK, Tisherman SA. Outcome and quality of life of patients with acute pancreatitis requiring intensive care. J Surg Res 2000; 91(1):89-94. doi: 10. 1006/jsre.2000.5925.
- Mutch KL, Heidal KB, Gross KH, Bertrand B. Cost-analysis of nutrition support in patients with severe acute pancreatitis. Int J Health Care Qual Assur 2011; 24(7):540-7. doi: 10. 1108/09526861111160571.
- Mole DJ, Gungabissoon U, Johnston P, Cochrane L, Hopkins L, Wyper GMA, et al. Identifying risk factors for progression to critical care admission and death among individuals with acute pancreatitis: a record linkage analysis of Scottish healthcare databases. BMJ Open 2016; 6(6):e011474. doi: 10.1136/bmjopen-2016-011474.
- Van Santvoort HC, Besselink MG, Bakker OJ, Hofker HS, Boermeester MA, Dejonget CH, et al. Dutch pancreatitis study group. A step-up approach or open necrosectomy for necrotizing pancreatitis. N Engl J Med 2010; 362(16): 1491-502. doi: 10.1056/NEJMoa0908821.
- Bakker OJ, van Santvoort HC, van Brunschot S, Geskus RB, Besselink MG, Bollen TL, et al. Dutch pancreatitis study group. Endoscopic transgastric vs surgical necrosectomy for infected necrotizing pancreatitis: A randomised trial. JAMA 2012; 307(10):1053-61. doi: 10.1001/jama.2012.276.
- Van Brunschot S, van Grinsven J, van Santvoort HC, Bakker OJ, Besselink MG, Boermeester MA, et al. Dutch pancreatitis study group. Endoscopic or surgical step-up approach for infected necrotising pancreatitis: A multicentre randomised trial. Lancet 2018; 391(10115):51-8. doi: 10.1016/S0140- 6736(17)32404-2.
- Moggia E, Koti R, Belgaumkar AP, Fazio F, Pereira SP, Davidson BR, et al. Pharmacological interventions for acute pancreatitis. Cochrane Database Syst Rev 2017; 4(4): CD011384. doi: 10.1002/14651858.CD011384.pub2
- Vivian E, Cler L, Conwell D, Coté GA, Dickerman R, Freeman M, et al. Acute pancreatitis task force on quality: Development of quality indicators for acute pancreatitis management. Am J Gastroenterol 2019; 114(8):1322-42. doi: 10. 14309/ajg.0000000000000264.
- Hines OJ, Pandol SJ. Management of severe acute pancreatitis. BMJ 2019; 367: l6227. doi: 10.1136/bmj.l6227.