Leiomyosarcoma of Inferior Vena Cava in an Immunocompetent Young-adult Female Patient
By Joao Filipe Gomes1, Ines Vieira2, Jorge Mendes1, David Donaire1, Rui Almeida3Affiliations
doi: 10.29271/jcpsp.2022.10.1353ABSTRACT
Leiomyosarcoma of the inferior vena cava is an extremely rare malignancy originating from the smooth muscle of the vessel wall, with only a few hundred cases reported in the literature. There are no clear guidelines for treatment, but surgical resection is currently the only curative option. Further research is needed to better understand the disease and guide its management.
We report a case of a 39-year woman who presented to the emergency room with a four-day history of nonspecific abdominal pain, nausea and fever. An abdominal computed tomography revealed a mass in relation with the inferior vena cava and hepatic nodules. Histological examination proved it to be a leiomyosarcoma of inferior vena cava with liver metastases.
Key Words: Leiomyosarcoma, Inferior vena cava, Sarcoma.
INTRODUCTION
Venous leiomyosarcomas account for 1 in every 100,000 malignant tumours, with leiomyosarcoma of inferior vena cava (LIVC) being responsible for more than half of these sarcomas.1 Primary LIVC is an extremely rare mesenchymal tumour originating from the smooth muscle of the vein wall (1) with a few hundred cases reported in the literature since its first description in 1871 by Perl.1-3
LIVC is more frequent in females (3.5:1) in their sixth decade of life.2,3 The incidence of smooth muscle tumours is higher in immunosuppressed patients.2
Due to its retroperitoneal location, the clinical presentation is usually nonspecific and has an insidious onset, all of which contribute to the latency of diagnosis, with 10% of all cases diagnosed incidentally.2,3 The most common presenting symptoms include nonspecific abdominal pain, lower-extremity oedema, and weight loss. Atypical presentations include atrial fibrillation and fever.1,2
CASE REPORT
A 39-year Caucasian woman with a recent diagnosis of hypertension was admitted to the emergency room with a four-day history of nonspecific abdominal pain, nausea and fever. Vital signs showed an auricular temperature of 39.1oC, normal blood pressure, pulse of 88 beats per minute, and respiratory rate of 20 cycles per minute. Physical examination revealed abdominal tenderness in the right hypochondrium with no further relevant abnormalities found.
Laboratory tests showed just mildly elevated C-reactive protein (7.08 mg/d; normal range: <0.5 mg/dL) and lactate dehydrogenase (303 U/L; normal range: 125-220 U/L). Chest radiography was normal.
An abdominal ultrasound showed two hypoechoic nodules located in the left hepatic lobe, measuring 3 and 3.6 cm; it also revealed a nodule adjacent to the vena cava suggestive of adenopathy with a 3 cm long axis.
The patient was admitted to the internal medicine ward since it was not possible to eliminate the suspicion of hepatic abscess. As the patient became apyretic on the second day after admission, the initial suspicion became less likely. Therefore, an abdominal computed tomography (CT) was performed and revealed an expansive lesion in the infra-renal vena cava, hypodense and heterogeneous, with both exophytic and endoluminal growth. The lesion emerged immediately below the renal veins, at a 5 mm distance from the right one, apparently without invading them. The radiologist considered this lesion likely to correspond to an LIVC (Figure 1). In the liver, two nodular formations were apparent: one of them, located in segment II, was heterogeneous and hypovascular, and measured around 28 mm, suggestive of metastasis; the other nodule was also located in segment II and was suggestive of focal nodular hyperplasia.
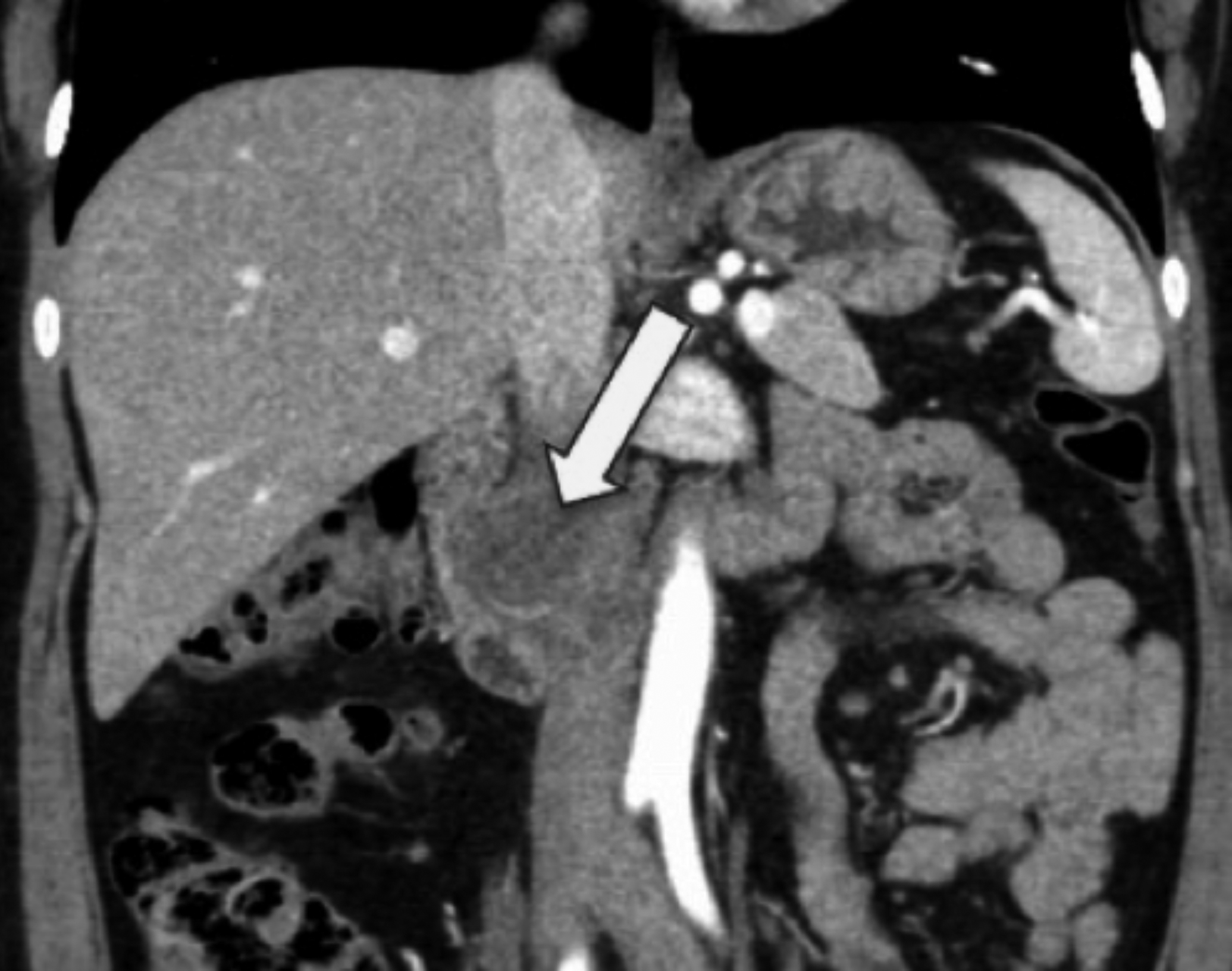 Figure 1: Computed tomography scan, sagittal cut, revealing a leiomyosarcoma of the inferior vena cava: expansive lesion in the infra-renal inferior vena cava, hypodense and heterogeneous, with endoluminal and also exophytic growth, measuring approximately 3.9 ×2.4 × 3.4 cm (T×AP×L).
Figure 1: Computed tomography scan, sagittal cut, revealing a leiomyosarcoma of the inferior vena cava: expansive lesion in the infra-renal inferior vena cava, hypodense and heterogeneous, with endoluminal and also exophytic growth, measuring approximately 3.9 ×2.4 × 3.4 cm (T×AP×L).
Meanwhile, the case was discussed with the surgical team and additional examinations for staging purposes were requested, namely a positron emission tomography (PET)-scan which revealed an intensely hypermetabolic lesion in the IVC, compatible with high-grade malignant neoplastic lesion with hepatic metastases.
Exploratory laparotomy did not reveal ascites and/or peritoneal implants. An intra-operative ultrasound identified two lesions in segment II of the liver. Resection of the involved segment of vena cava was performed with prosthetic replacement. Hepatic segments II and III were also removed. The patient had an uncomplicated postoperative course and was discharged four days after surgery.
The macroscopic examination revealed a 5.3 cm long vascular segment, surrounded by a nodular 5.5 × 3.5 × 3 cm tumour mass. The lesion was white/yellowish and firm with a multinodular and fasciculate pattern on sectioning. It was located in the vein wall, apparently under the conserved intima, leading to a decrease in the lumen and extending, on the periphery, to the surrounding adipose tissue. The liver lobectomy specimen showed two nodular lesions measuring 4 × 3.5 × 3 cm and 1.5 × 2 × 2 cm, separated by 1.2 and 4.5 cm from the surgical margins, respectively. The microscopy revealed a tumour mass composed of a fusocellular proliferation, densely cellular, with fasciculate pattern (Figure 2). There were hypocellular and hyalinised zones and extensive areas of necrosis (> 50% of the neoplasia - score 2). The lesion showed marked cytonuclear pleomorphism (score 3), absence of vascular invasion, and 60 mitoses/10 HPF/3mm2 (score 3), making a total score of 8 - G3 (National Federation of Fight Centers against Cancer / FNCLCC), with free circumferential margin and microscopic involvement of an inferior vena cava (IVC) resection margin - R1. Seven peri-tumoral and one of the hepatic hilar lymph nodes were examined, all without metastatic disease. Metastatic liver disease and focal nodular hyperplasia were present.
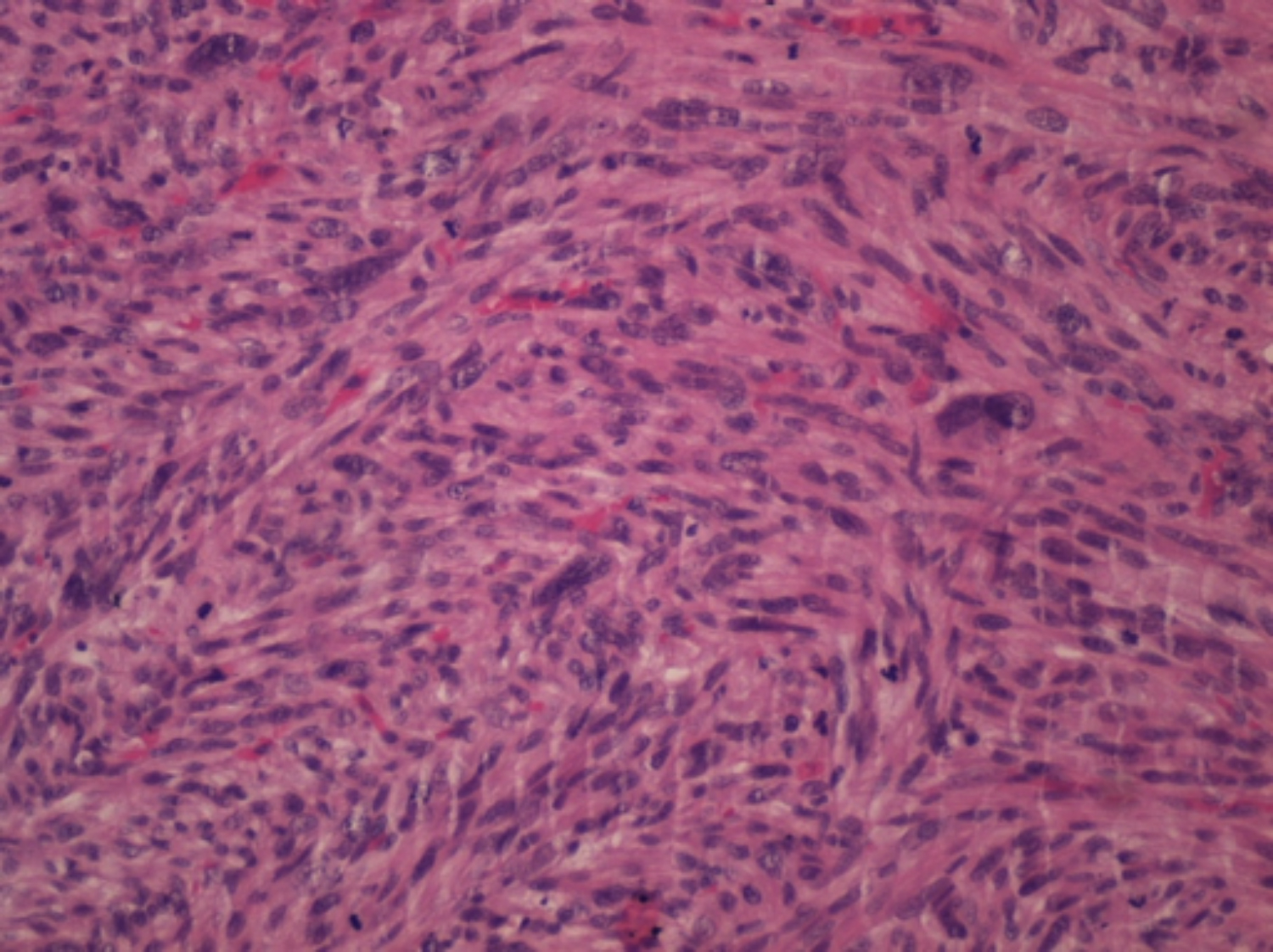 Figure 2: Histological section of leiomyosarcoma, stained with hematoxylin and eosin (H&E) revealing bundles of spindle cells with eosinophilic cytoplasm, cigar-shaped nuclei with severe atypia. (H&E, ×200).
Figure 2: Histological section of leiomyosarcoma, stained with hematoxylin and eosin (H&E) revealing bundles of spindle cells with eosinophilic cytoplasm, cigar-shaped nuclei with severe atypia. (H&E, ×200).
The patient underwent adjuvant chemotherapy with vincristine, Adriamycin, and cyclophosphamide regime (last cycle in 2018).
She is presently disease-free two years after her surgery with unremarkable laboratory parameters.
DISCUSSION
Despite being rare, LIVCs are the most frequent primary malignant tumours of the venous system.2
This tumour has a slow-growth, and it is weakly invasive with late metastatic spread, usually via a systemic route (liver, lung). Lymphatic spread is rare and usually late.4 Our patient corresponded to this pattern having presented as T2 N0 M1 tumour.
These tumours are classified into three groups according to the IVC segment involved: segment I (lower segment, from the iliac veins up to the renal veins); segment II (middle segment, from the renal veins to the hepatic confluence) and segment III (upper segment, from the hepatic confluence to the right atrium).1-3,5,6 Segment II LIVCs are the most common ones and can cause right upper quadrant abdominal pain, renovascular hypertension and, if there is occlusion, nephrotic syndrome may emerge. Segment III LIVCs may lead to Budd-Chiari syndrome with symptoms including abdominal distention, nausea, vomiting, hepatomegaly, ascites, and jaundice.2,5
Imaging studies are essential for the diagnosis, with computed tomography (CT) and magnetic resonance imaging (MRI) being the most commonly used.2 A hypodense retroperitoneal mass involving IVC with heterogeneous postcontrast enhancement is commonly described in CT studies.7 In the differential diagnosis, several lesions potentially involving the IVC should be considered, such as lymphoma, neurofibroma, liposarcoma, schwannoma, ganglioneuroma, paraganglioma, and teratoma. Hematoma, thrombus, and retroperitoneal fibrosis may also give rise to abnormalities in IVC imaging.8 In this case, the presence of fever and hepatic nodules led us to consider a hepatic abscess. This possibility was rapidly dismissed and, since the CT was very suggestive of a LIVC, this soon became our main diagnostic hypothesis.
Surgical resection is currently the only potential curative therapy.1,9 The role of adjuvant chemotherapy or radiotherapy is controversial.1-3 Older age, larger tumour size, en bloc resection, resection with positive margins and use of adjuvant chemotherapy were associated with worse prognosis.10 Presentation also influences prognosis with deep venous thrombosis, Budd-Chiari syndrome and segment III LIVC reflecting a poorer prognosis.6
This patient had a segment I LIVC, which is not the most common one. Her presentation was both typical, with abdominal pain, and atypical, with fever. She did not manifest lower extremity oedema. The patient had, however, been recently diagnosed with hypertension, which might have been related to renal vein involvement. She was 20 years younger than the usual age of presentation, which is in the sixth decade of life.2,3 As previously stated, these lesions are more common in immunosuppressed patients,2 which was not the case in our patient. Her young age, presentation without deep venous thrombosis and timely surgical intervention favoured her prognosis. The fact that microscopically positive margins were found may be a worrisome prognostic factor, reinforcing the need for long-term surveillance.
PATIENT’S CONSENT:
Informed consent was obtained from patients to publish the data concerning this case.
COMPETING INTEREST:
The authors declared no competing interest.
AUTHORS’ CONTRIBUTION:
JFG, IV: Produced the report and performed the literature review.
JM, DD: Assisted in the production of the report and the literature review.
RA: Oversaw the creation of the report and was involved in the care of the patient.
All the authors have approved the final version of the manuscript to be published.
REFERENCES
- Alkhalili E, Greenbaum A, Langsfeld M, Marek J, Rana MA, Glew R, et al. Leiomyosarcoma of the Inferior Vena Cava: A Case Series and Review of the Literature. Ann Vasc Surg 2016; 33:245-51. doi: 10.1016/j.avsg.2015.10.016.
- Xu J, Velayati A, Berger BJ, Liu M, Cheedella NKS, Gotlieb V. Leiomyosarcoma of the inferior vena cava in an HIV-positive adult patient: A case report and review of the literature. Am J Case Rep 2017; 18:1160-5. doi: 10.12659/ ajcr.905787.
- Gafarli S, Igna D, Wagner M, Nistor A, Glanemann M, Stange B. Dyspnea due to an uncommon vascular tumor: Leiomyosarcoma of the infrahepatic vena cava inferior. Surg Case Rep 2020; 6(1). doi: 10.1186/s40792-020- 00896-9.
- Sulpice L, Rayar M, Levi Sandri G-B, de Wailly P, Henno S, Turner K, et al. Leiomyosarcoma of the inferior vena cava. J Visc Surg 2016; 153(3):161-5. doi: 10.1016/j.jviscsurg. 2015.11.002.
- Keller K, Jacobi B, Jabal M, Stavrou GA. Leiomyosarcoma of the inferior vena cava: A case report of a rare tumor entity. Int J Surg Case Rep 2020; 71:50-3. doi: 10.1016/j.ijscr. 2020.04.094.
- Arif SH, Mohammed AA. Leiomyosarcoma of the inferior vena cava presenting as deep venous thrombosis; case report. Radiol Case Rep 2020; 15(2):133-5. doi: 10.1016/j. radcr.2019.10.034.
- Naphade PS, Raut AA, Hira P, Vaideeswar P, Vadeyar H. Leiomyosarcoma of the inferior vena cava. Arch Iran Med 2014; 17(5):383-7.
- Marko J, Wolfman D. Retroperitoneal leiomyosarcoma from the radiologic. Radiographics 2018; 38(5):1403-20. doi: 10.1148/rg.2018180006.
- Flores L, Ferrer J, Pages M, Ramírez J, Fuster J, García-Valdecasas JC. Leiomyosarcoma of the inferior vena cava: feasibility of surgical resection. A report of two cases. Rev Esp Enfermedades Dig 2015; 107(7):458-60.
- Wachtel H, Gupta M, Bartlett EK, Jackson BM, Kelz RR, Karakousis GC, et al. Outcomes after resection of leiomyosarcomas of the inferior vena cava: A pooled data analysis of 377 cases. Surg Oncol; 24(1):21-7. doi: 10. 1016/j.suronc.2014.10.007.