Comparison of Femoral and Internal Jugular Vein Catheterisation Trends in Sick Infants
By Ali Erdal Karakaya1, Ahmet Burak Dogan2, Ahmet Gokhan Guler2Affiliations
doi: 10.29271/jcpsp.2022.06.794ABSTRACT
Objective: To compare two different ways of central venous access in newborns regarding complications and success rates.
Study Design: Descriptive study.
Place and Duration of Study: Third-level Newborn Intensive Care Units in Kahramanmaras Sutçu Imam University Health Practice and Research Hospital and Megapark Private Hospital, Turkey, between July 2017 and May 2019.
Methodology: The data of 132 and 81 patients who underwent tunnelled femoral vein (FV) and percutaneous internal jugular vein (IJV) access procedures, respectively, for advanced medical management were reviewed. Planned procedures performed in an operating room under general anaesthesia were included in the study. Demographic data of infants, kind and the number of complications, and findings in clinical follow-up were recorded.
Results: There were similar success rates between techniques. No complication requiring intervention occurred during the placement of the FV catheters. Four procedures were interrupted due to periprocedural complications requiring intervention in the placement of IJV catheters. The median value of catheter duration was 25.5 (15-36.75) and 14 (9-20) days in FV and IJV group, respectively, and the difference is significant (p<0.001). Lower infectious complications (p=0.008) were detected in the use of FV catheters.
Conclusions: Similar success rates were found for both ways of central venous access. IJV stent's intrathoracic complications can be too severe for sick infants to cope with. FV stents can also be used in infants with well-tolerated complications. Further studies should confirm the low infectious complication rate of this study in FV catheters.
Key Words: Femoral vein, Internal jugular vein, Central venous access, Catheter-related infection, Infants.
INTRODUCTION
Central venous catheters (CVCs) have been a significant part of newborn care with the ability for long-term survival of sick and immature infants in neonatal intensive care units (NICU).1 Most premature and critically ill infants need a central line for advanced medical management and detailed monitoring in the further stages of their intensive care process. Nowadays, even in extremely low birth weight infants, central venous access under sonographic guidance can be performed safely with a high success rate.2 Nevertheless, the central thoracic vein access raises concern for complications related to intrathoracic vital organs.3
Significant complications involving lungs, heart, and great vessels may be difficult to cope with, especially in newborns striving to survive. Femoral vein (FV) access is free from the complications mentioned above. However, it carries the risk of infection and line sepsis due to the access site's proximity to the anogenital region.4,5 Besides, thrombotic and vaso-occlusive events of the lower extremity secondary to femoral vein access place considerable stress on practitioners and caregivers, although most are temporary.6
Studies comparing femoral and jugular vein stent placement and user experience in newborns are limited.5
The aim of the study was to compare outcomes of central venous catheters, which were placed under sonographic guidance into the FV and internal jugular vein (IJV), on sick infants admitted under intensive care.
METHODOLOGY
Data of infants who underwent FV and IJV catheterisation under sonographic guidance in ‘Kahramanmaraş Sutcu Imam University Health Practice’ and ‘Research Hospital and Megapark Private Hospital’ between July 2017 and May 2019 was reviewed retrospectively after the Clinical Investigations Ethical Committee's approval. All planned procedures performed in an operating room under general anaesthesia were included in the study. Infants who were critically ill and needed emergent venous access and all the second and third catheterisation procedures were excluded.
Finally, 81 infants who underwent percutaneous IJV access under sonographic guidance in Sutcu Imam University Health Practice and Research Hospital constituted the IJV group, and 132 infants who underwent femoral vein access under sonographic guidance in Megapark Private Hospital constituted the FV group.
In both the institutes, umbilical and peripheral venous access was preferred as the primary way of intravenous medication of newborns. Epicutaneo-caval catheters were the next choice. If there were no intact umbilical and peripheral veins, the central veins were attempted for catheterisation as the last choice. All the procedures were performed under general anaesthesia by pediatric surgeons. Patients were stented with a 22 G size-10 cm length (B. BRAUN Certofix Mono Paed S 110, Germany) central venous catheter kit set.
In Sutcu Imam University Health Practice and Research Hospital, right internal jugular vein access is preferred for advanced medical treatment. After general anaesthesia was administered in these procedures, the patient was laid supine and Trendelenburg position was obtained on the operating table, and a towel roll was placed under the shoulders. Then, the patient's head was turned about 45 degrees towards the opposite of the target IJV. After providing sterile conditions, IVJ was punctured under real-time sonographic guidance (GE Vivid S5 BT12 Cardiovascular Ultrasound and 4-13 MHz linear transducer). Once venous flashback was seen, the stent was cut to half-length and introduced into the vein over the guidewire (Seldinger technique).
Physicians at the Private Hospital of Megapark prefer femoral vein access to manage sick newborns based on their personal experience. In femoral vein catheterization, general anaesthesia is administered, and the patient is positioned supine on the operating table. Then the sterile condition is obtained. Under real-time sonographic guidance (Mindray DC-7 Diagnostic Ultrasound System and 4-10 MHz linear probe) target vein is viewed, and the groin skin is punctured. Once the venous flashback was seen by accessing the femoral vein, the Seldinger technique is used. Then the stent is introduced into the vein over the guidewire.
In the FV group, the catheter tip position was confirmed with a babygram in the operating room (Figure 1), with sonography in the IJV group. All the catheters are fixed to the skin with an adhesive-based stabiliser, including an in-stent kit and extra transparent adhesive tapes. The dressing was changed twice a week. Success is considered to achieve a functional catheter that is placed appropriately in a central vein without any complications (or with acceptable minor complications) in this study. Unless the catheter tip culture and blood culture indicate the same microorganism in the presence of clinical findings of sepsis, the current infection has not been considered catheter-related sepsis.7
Data were analysed using SPSS software version 18.0 (SPSS Inc., Chicago, IL, USA). Categorical data were given as frequencies and percentages, while quantitative data were expressed as median (IQR: 25th percentile-75th percentile. Demographics and catheter duration of the groups were compared with the Mann-Whitney U-test. The categorical data of groups were assessed with the chi-square test. A p-value <0.05 was considered significant.
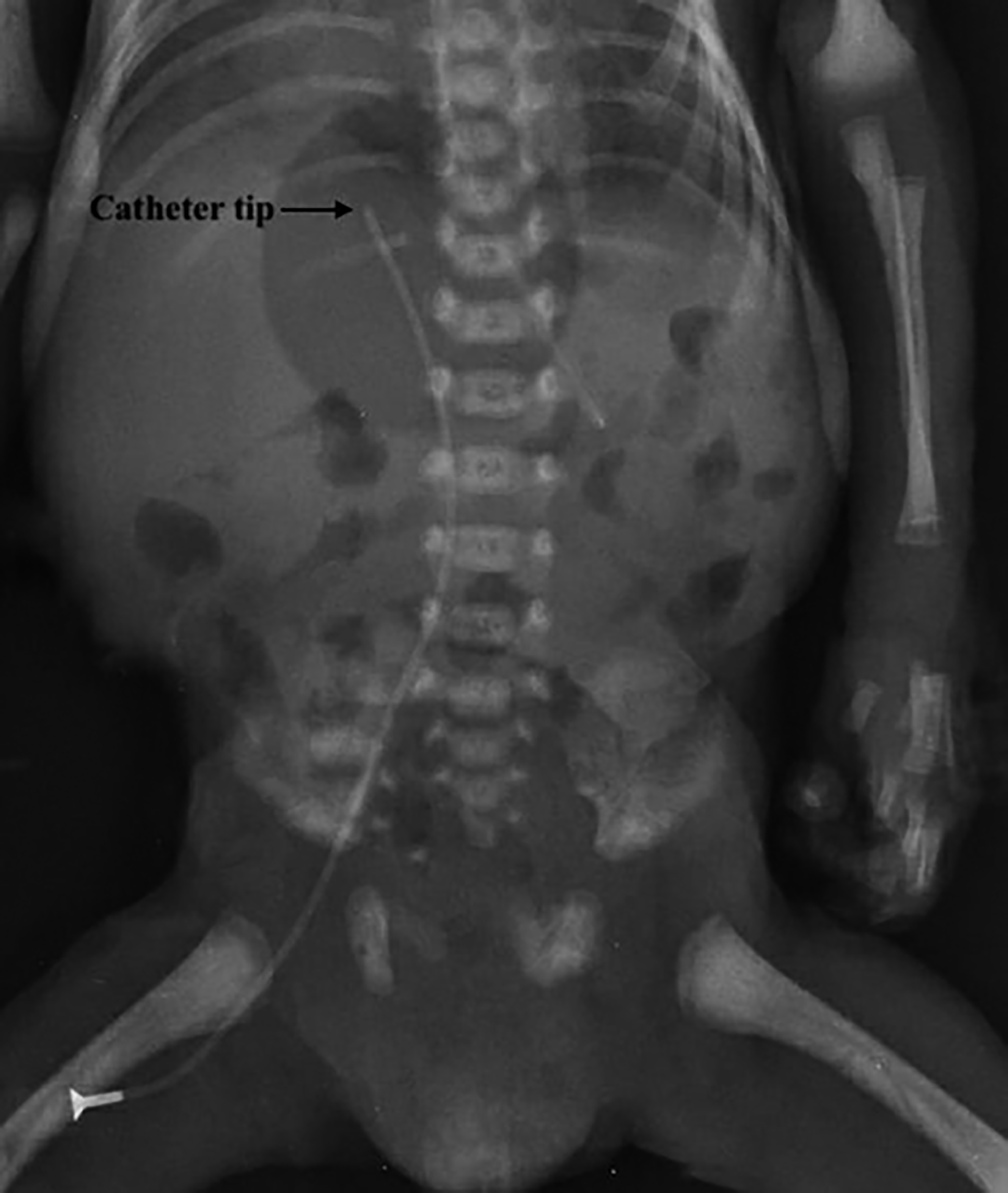 Figure 1: Catheter positioning with babygram.
Figure 1: Catheter positioning with babygram.
RESULTS
This study comprised the data of 213 newborns who underwent central venous access for advanced medical treatment. The median gestational age, birth weight, and age and weight at the procedure time are similar. The demographic data of infants were stated in Table I. In the FV group, 128 (97%) of 132 infants successfully catheterised. In the remaining four (3.0%), access to both FV failed because the guidewire could not be advanced through the vein. The left and right femoral vein catheterisations were achieved in 48 (36.4%) and 84 (63.6%) patients. The IJV group consisted of 81 infants, and eventually, 77 (95.1%) of them had success. Remainder four (4.9%) experienced periprocedural complications requiring immediate intervention. The primary admission diagnosis of the infants was given in Table I.
All the procedures in the IJV group were attempted on the right IJV. Since it was not known the number of vein access attempts for a successful catheterisation in a patient, information about the corrected success rates could not be estimated.
Table I: Demographics and admission diagnosis of the patients.|
|
FV (n:132) |
IJV (n:81) |
p |
|
Gender, m (%)/f (%) |
63(47.7) /69(52.3) |
47(58)/34(42) |
0.144 |
|
Gestational age, week (IQR) |
36 (32-38) |
36 (34-38) |
0.996 |
|
Birth weight, gram (IQR) |
2570 (1670-3200) |
2800 (2170-3210) |
0.055 |
|
Age at the time of the procedure, day (IQR) |
20 (15-30) |
19 (14-25) |
0.247 |
|
Weight at the time of the procedure, gram (IQR) |
2925 (1912.5-3300) |
2740 (2202.5-3250) |
0.767 |
|
Prematurity |
24 (18.2) |
18 (22.2) |
0.315 |
|
Sepsis |
22 (16.7) |
8 (9.9) |
|
|
IUGR |
7 (5.3) |
10(12.3) |
|
|
Feeding intolerance |
14 (10.6) |
8 (9.9) |
|
|
Surgical reasons |
13 (9.8) |
5 (6) |
|
|
RDS |
18 (13.6) |
12 (14.8) |
|
|
Congenital heart disease |
12 (9.1) |
4 (4.9) |
|
|
Central nervous system anomalies |
4 (3) |
5 (6.2) |
|
|
Asphyxia |
13 (9.8) |
5 (6.2) |
|
|
Other diagnoses |
5 (3.8) |
6 (7.4) |
|
|
FV: Femoral vein, IJV: Internal jugular vein; m/f: Male / Female; IQR: Interquartile range. |
|||
Table II: Comparison of groups.
|
|
FV (n=132) |
IJV (n=81) |
p |
|
Overall success rates, n (%) |
128(97) |
77(95.1) |
0.483 |
|
Overall complication rates, n (%) |
22(16.7) |
23(28.4) |
0.042 |
|
Catheter duration, day (IQR) |
25.5 (15-36.75) |
14 (9-20) |
<0.001 |
|
Procedural complications that required intervention |
>0.999 |
||
|
Pneumothorax* |
0 |
2 (2.5) |
|
|
Hemothorax* |
0 |
1 (1.2) |
|
|
Pleural dislodgement* |
0 |
1 (1.2) |
|
|
Procedural complications that did not require intervention |
|||
|
Hematoma |
3 (2.3) |
1 (1.2) |
|
|
Arterial catheterization |
0 |
1 (1.2) |
|
|
Transient extremity engorgement |
5 (3.8) |
0 |
|
|
Transient extremity cyanosis |
3 (2.3) |
0 |
|
|
Postprocedural infectious complications |
0.008 |
||
|
Catheter-related sepsis |
0 |
4 (4.9) |
|
|
Likely catheter-related sepsis |
5 (3.8) |
3 (3.7) |
|
|
Exit site infection |
6 (4.5) |
10 (12.3) |
|
|
FV: Femoral vein, IJV: Internal jugular vein; IQR: Interquartile range; * The reason for access failure. |
|||
When the overall success rates were compared (Table II), no significant difference was found between the two groups (p=0.483). Total catheter-days were 3995 and 1219 in FV and IJV groups, respectively. The median value of catheter duration was 25.5 (15-36.75) and 14 (9-20) days in FV and IJV groups, respectively, and the difference is significant, as well (p<0.001).
The most common reason for catheter removal in both groups was the end of treatment. However, a higher number of catheters (113 [85.6%]) could be used until the end of treatment in the FV group than in the IJV group (51 [63%]) (p<0.001).
Periprocedural and early postprocedural complications are listed in Table II. There was no complication requiring intervention during FV access. Complication involving the artery adjacent to the target vein was only seen in the IJV group. Although eight (6.1%) patients in the FV group had engorgement or discolouration of the lower limbs, doppler sonography ruled out thrombosis. The groups are similar in terms of periprocedural and early postprocedural complication rates (p>0.999).
In the late postprocedural period (Table II), catheter-related sepsis was detected only in four (4.9%) patients in the IJV group. Stent tip and blood cultures indicated Staphylococcus epidermidis (n = 1), Staphylococcus aureus (n = 2) and Candida albicans (n = 1). In both groups, the clinical findings of the sepsis regressed within 48-hour of stent removal in eight patients who were considered catheter-related sepsis. Still, stent tip cultures did not indicate any microorganisms. A total of 16 patients with stent exit-site infections were medically treated without stent removal in both groups. Postoperative infectious complication rates were significantly higher in the IJV group than in the FV group (p = 0.008).
DISCUSSION
In this study, the overall success rates were determined by 97% in the FV group and 95.1% in the IJV group (p= 0.483). The procedure failed in four (3%) patients in the FV group. Although the vein access was confirmed sonographically in these infants, the guidewire could not be advanced through the femoral vein. This may be because a large peripheral vein was attempted to be catheterized instead of the femoral vein. In these cases, the guidewire must have been directed to other minor veins instead of the femoral vein. Bruzoni et al. stated a similar problem with the standard guidewire in the sonographic-guided internal jugular vein access, and they remedied the problem with a micro-puncture introducer set.8 However, they did not suggest any reason for this condition. In the IJV group, four (4.9%) patients had failed catheterisation attempts due to periprocedural complications (pneumothorax, hemothorax, and pleural dislodgement of the stent). These procedures were terminated for urgent interventions. The overall success rates in both groups are comparable with the recent sonographic guided central venous access studies in the literature reporting overall success rates of between 92.3% and 100%.8-11
The first attempt accomplishment rate of CVC could not be obtained in the retrospective analysis of the data.
Although sonographic guidance increases CVC's safety, the procedure's mechanical complications may occur depending on patient age and practitioner experiences.12 These complications may be fatal in percutaneous access to central thoracic veins and may require lifesaving interventions and further increase the morbidity of the infants who are already treated in the NICU.3 The authors had to perform tube thoracostomy in four patients due to the pneumothorax (n=2), hemothorax (n=1), and intrapleural dislodgment of the catheter tip (n=1). The authors think that the rates of these mechanical complications will decrease with the increase of sonographic guidance experience in CVC placement on premature infants. However, any precaution, assistive technique, and experience level cannot eliminate all these central thoracic vein access complications.13
On the other hand, there is less concern for such fatal complications in FV access. There was only reported accidental femoral artery puncture in an old publication at the rates of 18% as a periprocedural complication during landmark guided femoral vein catheterisation.14 In recent studies on catheterisation of the femoral vein under sonographic guidance, no periprocedural complication other than transient extremity swelling (related to thrombosis or not), which is rare (1.1%), has been reported.1,5,9 The complications in the FV group are consistent with these literature data. However, despite similar extremity engorgement and discolouration rates, doppler USG confirmed the FV patency without thrombosis. All swelling and discolouration resolved within 24-hour without any intervention and stent removal. This transient response of veins demonstrated radiologically may be related to catheter-induced venous spasms.6 The mechanism of the spasm is incompletely understood. The chemical effect caused by the needle or guidewire may be responsible.16 In the absence of thrombosis, close monitoring of extremity without removal may be a way of catheter management.
In this study, the overall complications during stent placement and use were more common in the IJV group than in the FV group. The difference is statistically significant (p=0.042). Postprocedural infectious complications were significantly higher in the IJV group than in the FV group (p=0.008), although catheter duration and total catheter days were significantly higher in the FV group. In the IJV group, four stents were removed, and the catheter tip culture-confirmed CRS. CRS was not demonstrated in any patients in the FV group. Eight stents (five and three stents in the IJV and FV group, respectively) were removed with suspected catheter-associated sepsis; however, any microorganism was not detected in catheter tip cultures. There is not enough data to show why CRS rates are high in the IJV group. However, in this group, it was a little challenging to maintain the IJV catheters that were cut to half clean and active in infants' short and flex necks. Especially in intubated infants without swallowing reflex, oral secretion, and body sweat complicated the stent fixation and wound dressing and may have contaminated the stents.
Actually, catheters placed in the groin area would have been expected to produce more infection complications due to faecal contamination. However, many studies indicated similar infectious complication rates between femoral and non-femoral catheters in children.17-19 Similar results have been reported in adult studies as well.20 CRS was not diagnosed at all in the FV group; however, it was considered possible in a total of five patients. The significantly lower rate of infectious complications in the FV group may be associated with a more stable catheter dressing (on the inguinofemoral region) and quality of catheter care. The current data are not at a level to demonstrate that FV catheters are superior to IJV catheters in terms of catheter-related infection. However, as a literature data, placement of the catheter hub away from the neck and groin with tunnelling or the cuffed and tunnelled catheters like Broviac would reduce the rate of infectious complications.15,21,22
With the current data of the study, FV access seems to be prominent with low complication rates. However, this study has several limitations, mainly its retrospective nature and the patient's limited number. Possible biases of two different clinics in central venous access management may have adversely affected the study results. More valuable results could be revealed if the late doppler assessment of attempted veins could be included in the study.
Further prospective studies with a sufficient number of patients about newborn central venous access would demonstrate more reliable results.
CONCLUSION
Similar and acceptable success rates were found for both ways of central venous access. Sonographic guidance significantly reduced the rate of IJV catheter placement complications consistent with the literature data; however, its’ intrathoracic complications can be too severe for sick infants to cope with. FV stents can be favourable because infants well tolerate them without any severe complications. Further studies should confirm the superiority of FV catheters observed over IJV catheters in infection complications.
ETHICAL APPROVAL:
The study was conducted after the Clinical Investigations Ethical Committee's approval.
PATIENTS’ CONSENT:
As the study was retrospective, the patients' consents were waived.
COMPETING INTEREST:
The authors declared no competing interest.
AUTHORS’ CONTRIBUTION:
AEK, ABD, AGG: Substantial contributions to conception and design, or analysis and interpretation of data.
AEK, AGG: Drafting the article or revising it critically for important intellectual content.
ABD: Final approval of the version to be published.
AEK, ABD, AGG: Agreement to be accountable for all aspects of the work.
REFERENCES
- Eichenwald EC. Extremely Low-Birth-Weight Infants. Tenth Edit. Elsevier Inc 2017. doi:10.1016/B978-0-323-40139-5. 00031-0
- Montes-Tapia F, Rodríguez-Taméz A, Hernandez-Garduño A, Barreto-Arroyo I, Rodríguez-Balderrama I, de la O-Cavazos M, et al. Vascular assessment of the right internal jugular vein in low birth weight newborns. J Matern Fetal Neonatal Med 2013; 26(15):1476-7058. doi:10.3109/14767058. 2013.789853.
- Eisen LA, Narasimhan M, Berger JS, Mayo PH, Rosen MJ, Schneider RF. Mechanical complications of central venous catheters. J Intensive Care Med 2006; 21(1):40-46. doi:10.1177/0885066605280884.
- Krishnamurthy G, Keller MS. Vascular access in children. Cardiovasc Intervent Radiol 2011; 34(1):14-24. doi:10. 1007/s00270-010-9865-3.
- Tsai MH, Lien R, Wang JW. Complication rates with central venous catheters inserted at femoral and non-femoral sites in very low birth weight infants. Pediatr Infect Dis J 2009; 28(11):966-70. doi:10.1097/INF.0b013e3181aa3a29.
- Clemens RK, Lillis AP, Alomari AI. Catheter-induced venous spasm. Circulation 2012; 126(19):2363-65. doi:10.1161/CIRCULATIONAHA.112.112946.
- Silberzweig JE, Sacks D, Khorsandi AS, Bakal CW. Reporting Standards for Central Venous Access. J Vasc Interv Radiol 2003; 14(9 PART 2):443-52. doi:10.1097/01.RVI.00000 94617.61428.bc.
- Bruzoni M, Slater BJ, Wall J, St Peter SD, Dutta S. A prospective randomised trial of ultrasound - Vs landmark-guided central venous access in the pediatric population. J Am Coll Surg 2013; 216(5):939-43. doi:10.1016/j.jamcollsurg.2013. 01.054.
- Gaballah M, Krishnamurthy G, Keller MS, McIntosh A, Munson DA, Cahill AM. US-guided placement and tip position confirmation for lower-extremity central venous access in neonates and infants with comparison versus conventional insertion. J Vasc Interv Radiol 2014; 25(4):548-55. doi:10.1016/j.jvir.2014.01.003.
- Montes-Tapia F, Rodríguez-Taméz A, Cura-Esquivel I. Efficacy and safety of ultrasound-guided internal jugular vein catheterisation in low birth weight newborn. J Pediatr Surg 2016; 51(10):1700-3. doi:10.1016/j.jpedsurg.2016.05.014.
- Lausten-Thomsen U, Merchaoui Z, Dubois C. Ultrasound-guided subclavian vein Cannulation in low birth weight neonates. Pediatr Crit Care Med 2017; 18(2):172-5. doi:10.1097/PCC.0000000000001028.
- Rivera-Tocancipá D, Díaz-Sánchez E, Montalvo-Arce CA. Ultrasound versus anatomical landmarks: Immediate complications in the central venous catheterization in children under 18 years of age. Rev Española Anestesioly Reanim (English Ed. 2018; 65(7):366-72. doi:10.1016/j.redare.2018. 02.014.
- Bauman M, Braude D, Crandall C. Ultrasound-guidance vs. standard technique in difficult vascular access patients by ED technicians. Am J Emerg Med 2009; 27(2):135-40. doi:10.1016/j.ajem.2008.02.005.
- Abdulla F, Dietrich KA, Pramanik AK. Percutaneous femoral venous catheterisation in preterm neonates. J Pediatr 1990; 117(5):788-91. doi:10.1016/S0022-3476(05)83346-3.
- Gaballah M, Krishnamurthy G, Berman JI. Lower extremity vascular access in neonates and infants: A single institutional experience. J Vasc Interv Radiol 2015; 26(11): 1660-68. doi:10.1016/j.jvir.2015.08.004.
- Duan X, Ling F, Shen Y, Yang J, Xu HY. Venous spasm during contrast-guided axillary vein puncture for pacemaker or defibrillator lead implantation. Europace 2012; 14(7):1008- 1011. doi:10.1093/europace/eus066.
- Haas NA. Clinical review: Vascular access for fluid infusion in children. Crit Care 2004; 8(6):478-84. doi:10.1186/cc2880.
- Karapınar B, Cura A. Complications of central venous catheterisation in critically ill children. Pediatr Int 2007; 49(5):593-9. doi:10.1111/j.1442-200X.2007.02407.x.
- Noonan PJ, Hanson SJ, Simpson PM, Dasgupta M, Petersen TL. Comparison of complication rates of central venous catheters versus peripherally inserted central venous catheters in pediatric patients. Pediatr Crit Care Med 2018; 19(12):1097-1105. doi:10.1097/PCC.0000000000001707.
- Marik PE, Flemmer M, Harrison W. The risk of catheter-related bloodstream infection with femoral venous catheters as compared to subclavian and internal jugular venous catheters: A systematic review of the literature and meta-analysis. Crit Care Med 2012; 40(8):2479-85. doi:10.1097/CCM.0b013e318255d9bc.
- Lindquester WS, Hawkins CM, Monroe EJ. Single-stick tunneled central venous access using the jugular veins in infants weighing less than 5 kg. Pediatr Radiol 2017; 47(12):1682-87. doi:10.1007/s00247-017-3937-3.
- Nahum E, Levy I, Katz J. Efficacy of subcutaneous tunneling for prevention of bacterial colonisation of femoral central venous catheters in critically ill children. Pediatr Infect Dis J 2002; 21(11):1000-4. doi:10.1097/00006454-200211000-00005.