A Young Man with a Rare Case of Adrenal Incidentaloma
By Usama Bin Khalid, Qurat Ul Ain, Zujaja Hina Haroon, Muhammad Aamir, Shakeel Ahmad, Shahrukh ShahAffiliations
doi: 10.29271/jcpsp.2021.12.1497ABSTRACT
Adrenal incidentalomas (AIs) are adrenal masses discovered as incidental finding, often on imaging studies, unrelated to adrenal disorders. Sometimes, they are discovered as part of work-up for adrenal pathology. AIs are mostly nonfunctional (hormonally silent), but can also be functional (hormonally active). The differential diagnosis includes many primary, metastatic, benign and malignant conditions.
The current case is a young male, who went to a peripheral health facility with complaints of dysuria and burning micturition. His laboratory and radiological investigations suggested a urinary tract infection with an incidental finding of adrenal mass. After referral, his detailed work-up was done at Endocrine Clinic of Armed Forces Institute of Pathology (AFIP), Rawalpindi, which revealed history of episodic headaches and palpitations with paroxysmal spikes of high blood pressure up to 200/120 mmHg. Adrenalectomy was performed by laparoscopic surgery. Histopathological examination confirmed the diagnosis of pheochromocytoma with PASS score of 5/20. This is one of the rare cases of adrenal incidentaloma, often an autopsy finding. Pheochromocytoma needs to be investigated in all cases of AIs.
Key Words: Adrenal incidentaloma, Pheochromocytoma, Urinary tract infection.
INTRODUCTION
Adrenal masses discovered incidentally are termed as adrenal incidentalomas (AIs). They are often discovered on imaging studies unrelated to adrenal pathology; and usually, the patient has no signs and symptoms of adrenal hormonal excess or obvious underlying malignancy.1 Less commonly, they are discovered as part of workup for adrenal disorders (e.g. Cushing’s, Pheochromocytoma etc.). The differential diagnosis of AIs includes many primary, metastatic, benign, and malignant entities arising from adrenal cortex or medulla. AIs are mostly non-functional (hormonally silent), while 15% cases are found to be functional, i.e., having active disease (e.g. pheochromocytoma, primary aldosteronism, Cushing's, adrenal carcinomas, etc.), which require further evaluation and treatment to avoid medical complications.2 Work-up for AIs must be started to rule out any functional disorder and is invariably started with evaluation of adrenal profile. Serum cortisol, corticotrophin, plasma aldosterone, and renin levels should be assessed.
Pheochromocytoma being one of the most common among AIs must be evaluated by measuring 24 hours urinary vanillyl mandelic acid (VMA) and plasma metanephrine and non-metanephrine levels. As adrenal gland is the fourth common site for metastasis, work-up must include ruling out cancers of other organs as well as parathyroid hormone levels and calcitonin for pheochromocytoma associated malignancy (MEN2A).
CASE REPORT
A 25-year male presented with complaints of dysuria and burning micturition for 5 days at a peripheral hospital. Initial imaging by ultrasound kidney ureter bladder (USG KUB) revealed an adrenal mass. He was treated for urinary tract infection (UTI) and was referred to a tertiary care hospital for further work-up. Detailed evaluation was done when he presented to the Endocrine Clinic of Armed Forces Institute of Pathology (AFIP), Rawalpindi.
Detailed history revealed complaints of recurrent headache and palpitations for last 2 years. Headache was intermittent, moderate to severe in intensity, throbbing in character and was associated with photophobia, phono-phobia and dizziness. Palpitations were episodic, lasting 5-10 minutes, recurring 3-4 times daily with more frequency at night, associated with heat intolerance, sweating and feeling of chest tightness. There was no blurring of vision or any neurological, respiratory and gastrointestinal system related complaints. General physical examination revealed a young man of average built, conscious and well oriented, having tachycardia and high blood pressure occurring as paroxysmal attacks going up to 200/120 mmHg. Electrocardiogram showed sinus tachycardia with left ventricular hypertrophy (LVH). Echocardiogram showed 60% ejection fraction and concentric LVH. Routine hematology and chemistry analysis was done which showed normal complete blood counts, liver function tests, renal function tests, serum electrolytes and plasma glucose. Thyroid functions were also normal with TSH 2.2 µIU/mL (normal: 0.4-4.5) and fT4 21.6 pmol/l (normal: 10.3-34.7). Imaging with contrast enhanced computerised tomography (CECT) with adrenal protocol was carried out for confirming initial USG finding of adrenal mass, which revealed adrenal mass, measuring 4.4×5.4×6.5 cm (Figure 1). Further work-up for AI was done to rule out adrenal insufficiency, hypersecretion or any metastasis. Plasma aldosterone and morning cortisol levels along with low dose dexamethasone suppression test and plasma active renin concentration (ARC) were found normal, i.e., 385 pmol/L (normal: 140-830), 399 nmol/L (normal: 138-690), 49.5 nmol/l (normal: <50nmol/l) and 30.5 µIU/mL (normal: 8-35), respectively. Tumor markers (AFP, CEA and CA 19-9) were evaluated for any underlying malignancy with adrenal metastasis, which were normal. Pheochromocytoma was suspected and 24 hours urinary vanillyl mandelic acid (VMA) levels were evaluated, which were surprisingly within normal physiological range i.e. 42 µmol/24hr (normal: 5-56). Free plasma metanephrine and nor-metanephrine levels were raised almost 10-folds, i.e., 5.3 nmol/L (normal: <0.5) and 8.3 nmol/L (normal: <0.9), respectively, leading to the diagnosis of pheochromocytoma. Metaiodobenzylguanidine (MIBG) scintiscan was done to look for any metastasis, which showed an avid lesion in left supra-renal region, ruling out any metastatic deposits elsewhere in the body. Multidisciplinary team approach was adopted which included surgeon, anesthetist, medical specialist and pathologist. Medical therapy with phenoxybenzamine followed by propranolol was started to relieve recurrent headaches, palpitations, and to prevent hypertensive crisis. Alprazolam was prescribed for anxiety symptoms. He responded well to the specific medical treatment. General surgery team performed his adrenalectomy laparoscopically. During surgery, the blood pressure was controlled with intravenous (IV) labetalol 20 mg bolus and infusion at 2 mg/minute along with IV phenylephrine and nor-adrenaline boluses on hypotensive episodes. A 8×7×5 cm left adrenal mass weighing 75 g with clear margins was resected and sent for histopathological review. Histopathological analysis revealed pheochromocytoma, having PASS score of 5/20. Follow-up showed normal post-operative plasma metanephrine 0.05 nmol/l (normal: 0.05-0.34) and nor-metanephrine levels 0.46 nmol/l (normal: 0.10-0.55), respectively.
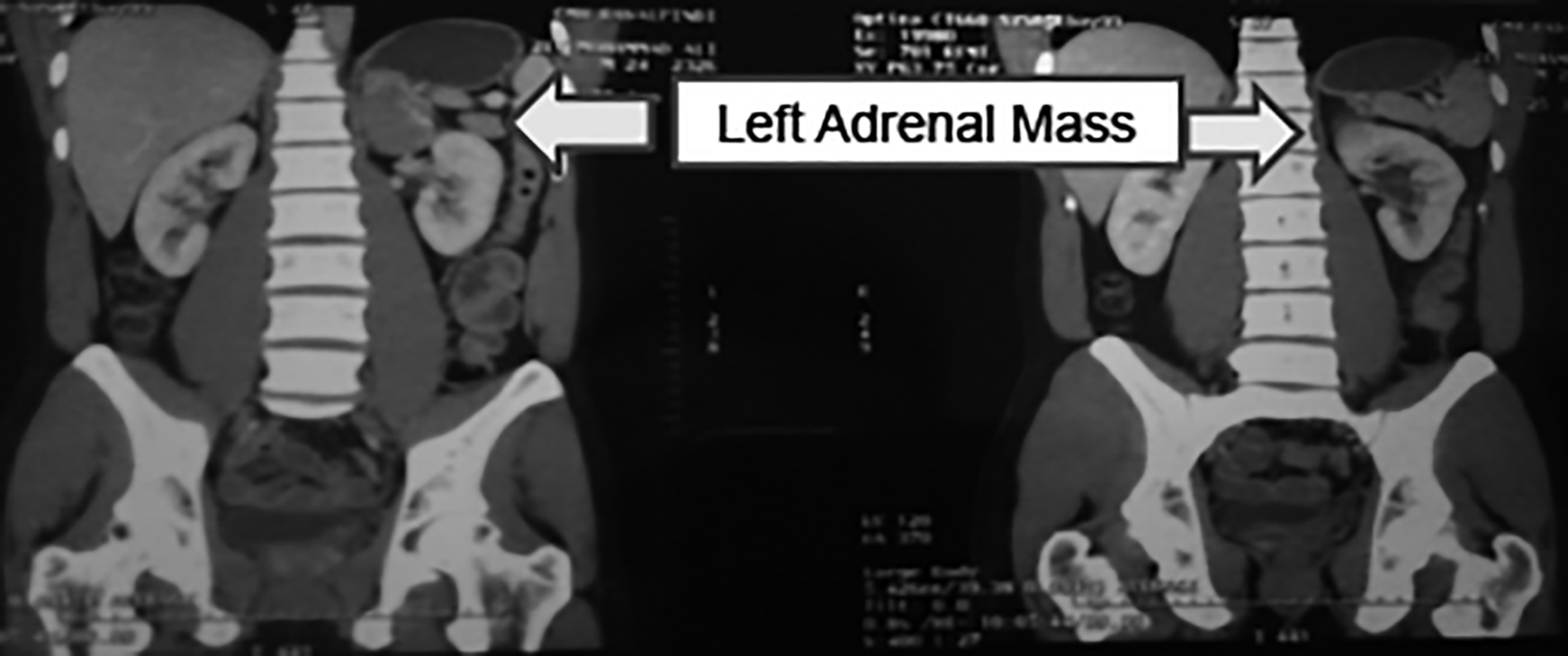 Figure 1: Contrast enhanced computerised tomography (CECT) of abdomen with adrenal protocol.
Figure 1: Contrast enhanced computerised tomography (CECT) of abdomen with adrenal protocol.
DISCUSSION
The case reported here is AI, diagnosed as pheochromocytoma. AIs are reported more nowadays, largely due to increased use of imaging modalities for clinical work-up of different diseases. The prevalence is about 10% in general population, while the autopsy prevalence for AIs is 2-9%.3 The prevalence of AIs increases with age with no gender predilection in adults; the rate is less than 1% in patients <30 years, and 7% in patients >70 years of age. AIs prevalence is higher in hypertensive, diabetic and obese persons.4 About 1-5% cases of AIs are diagnosed on the basis of CT scans obtained for reasons other than adrenal pathology.5 The most important hormonally silent adrenal incidentaloma is pheochromocytoma.6 The prevalence of pheochromocytoma is 1-10% of all AIs. A study by Falhammar et al. of 94 cases of pheochromocytoma determined that 64% of cases were identified incidentally, while 32% were found in patients suspected of having adrenal disorders. In another 4% of cases, patients were being screened for MEN2A.7
Work-up for AIs must be started to rule out any functional disorder. Adrenal insufficiency should be the first consideration, especially with bilateral AIs. Short synacthen (ACTH) test, a dynamic function test helps to rule out the diagnosis. Adrenal cortical adenoma can be diagnosed with a high degree of accuracy using imaging modalities. The adrenal gland is the fourth most common site of metastasis, and adrenal metastases may be found in as many as 25% of patients with known primary lesions. The treatment for a hormonally active (functional) adrenal tumor is surgery. The treatment for a malignancy depends on the cell type, spread, and location of the primary tumor. Nonfunctional adrenal cortical adenomas are not premalignant, and surgical excision is not indicated.8
The patients with pheochromocytoma need to be treated with care as they present with vague symptoms and are easily missed. Multidisciplinary approach must be adopted which includes medical specialist, surgeon, anesthetist and pathologist. The malignant potential must be thoroughly evaluated. PASS score ≥4 suggests a malignant potential.9
Pheochromocytoma should be considered in all AI cases because they are more common than previously thought, the diagnosis is often overlooked, and a failure to recognize them may lead to patient death.
CONFLICT OF INTEREST:
The authors declared no conflict of interest.
AUTHORS’ CONTRIBUTION:
UBK: Idea, data collection.
QA: Stastical analysis, data interpretation.
ZHH: Review and correction.
MA: Review and data interpretation.
SA: Discussion and preview.
SS: Literature review.
REFERENCES
- Terzolo M, Bovio S, Pia A, Reimondo G, Angeli A. Management of adrenal incidentaloma. Best Pract Res Clin Endocrinol Metab 2009; 23(2):233-43. doi.org/10.1016/j. beem.2009.04.001.
- Cawood TJ, Hunt PJ, O'Shea D, Cole D, Soule S. Recommended evaluation of adrenal incidentalomas is costly has high false-positive rates and confers a risk of fatal cancer that is similar to the risk of the adrenal lesion becoming malignant; time for a rethink? Eur J Endocrinol 2009; 161(4):513-27. doi: 10.1530/EJE-09-0234.
- Abecassis M, McLoughlin MJ, Langer B, Kudlow JE. Serendipitous adrenal masses: Prevalence, significance, and management. Am J Surg 1985; 149(6):783-8. doi: 10.1016/s0002-9610(85)80186-0.
- Terzolo M, Stigliano A, Chiodini I, Loli P, Furlani L, Arnaldi G. AME position statement on adrenal incidentaloma. Eur J Endocrinol 2011; 164(6):851-70. doi: 10.1530/EJE-10-1147.
- Bovio S, Cataldi A, Reimondo G, Sperone P, Novello S, Berruti A, et al. Prevalence of adrenal incidentaloma in a contemporary computerized tomography series. J Endocrinol Invest 2006; 29(4):298-302. doi: 10.1007/ BF03344099.
- Yener S, Ertilav S, Secil M, Demir T, Akinci B, Kebapcilar L, et al. Prospective evaluation of tumor size and hormonal status in adrenal incidentalomas. J Endocrinol Invest 2010; 33(1):32-6. doi: 10.1007/BF03346546.
- Falhammar H, Kjellman M, Calissendorff J. Initial clinical presentation and spectrum of pheochromocytoma: A study of 94 cases from a single center. Endocr Connect 2018; 7(1):186-92. doi: 10.1530/EC-17-0321.
- Zeiger MA, Thompson GB, Duh QY, Hamrahian AH, Angelos P, Elaraj D, et al. The American association of clinical endocrinologists and American association of endocrine surgeons medical guidelines for the management of adrenal incidentalomas. Endocr Pract 2009; 15(Suppl 1):1-20. doi: 10.4158/EP.15.S1.1.
- Thompson LD. Pheochromocytoma of the adrenal gland scaled score (pass) to separate benign from malignant neoplasms: A clinicopathologic and immunophenotypic study of 100 patients. Am J Surg Pathol 2002; 26(5): 551-66. doi: 10.1097/00000478-200205000-00002.